Climate is not the same thing as weather. Weather is the minute-by-minute variable condition of the atmosphere on a local scale. Climate is the expected yearly weather conditions established over decades. Jump to Climate is NOT Weather
Culture, Climate Science & Education
Click the Double Arrows () to Explore this Principle

Principle Four: Climate is Variable
The Cultural Value is Flexibility
Episode Four: First Food
Episode 4: First Food
Transcript with Description of Visuals
|
Audio |
Visual |
|---|---|
|
Soft instrumental music: |
Aerial view of the Flathead River in black and white. |
|
Voice Over continues: |
A historic black and white photo of a woman with two children: one, a toddler, stands next to her; the other, a baby, rests in a cradleboard. |
|
and the sun sends a spiritual bird to come down. |
Camera is flying down the river toward a setting sun. |
|
The bird gave her a blessing. Her silvery hair becomes a plant. Her tears of sadness goes into this root. |
A historic black and white photo of an old woman peeling bitterroot. Next to her is a burlap blanket covered with cleaned bitterroot. |
|
The silvery hair is the plant itself that grows near the ground, and becomes food for the people. |
A digging stick, sunk into the prairie earth, works the soil, gently lifting a clump that contains a bitterroot plant. A hand then works the clump, peeling the soil away from the roots of the bitterroot plant. |
|
(soft instrumental music) |
Alyssa Pretty On Top and her mother walk through sagebrush along a low prairie ridge. |
|
How each spring we welcome it, like we are welcoming a loved one we have not seen for a long time. Bitterroot is a visitor, it is only here for a short time each year, and so the feast is when we come together as a tribe to welcome it back. |
A digging stick is pushed into the soil to lift another bitterroot. Alyssa and another girl are crouched a few feet apart, each is working to clean the soil from a bitterroot plant. Alyssa places the cleaned root into a basket that hangs from her shoulder. Scene changes to the feast at the Longhouse, a large room, filled with people seated at tables. |
|
It is our first food celebration. Families do not dig Bitterroot or harvest any other plant for their own use until after we have the Bitterroot Feast. We never know exactly when the Bitterroot harvest and feast will be. |
Various scenes from around the room. Cultural leader speaking, people at tables listening, people serving themselves with food from large bowls. |
|
The plant has to be ready for us to welcome it. So beginning in April, the elders and others who know Bitterroot observe it, watching for subtle changes in its leaves. Bitterroot tells us when it is ready. The harvest and feast has taught me some important things. First, how important it is to be observant of the natural world. In our culture, to be observant means to be fully present, fully engaged, enough to know and understand the plants and animals that feed and help us. |
Bitterroot harvest. Lots of people gathered, many with digging sticks. Transition to various scenes small and large groups of people and individuals digging bitterroot. |
|
Bitterroot tells us when it is ready. That requires knowing the plant as one knows a loved one. |
Child cleaning bitterroot roots. |
|
The second thing I have learned is the importance of flexibility. |
Mother working with two young children, teaching them how to clean bitterroot. |
|
Because the climate is changing, getting warmer, the time for the Bitterroot Feast has been changing. |
Woman carefully and lovingly cleaning bitterroot. |
|
In some years, it has been much earlier than ever before. |
Woman showing child how to clean roots and rebury heart of the plant. |
|
But we are adaptable and resilient. We can change, just as our ancestors have so many times in the past. |
Child looking intently at plant for the heart. |
|
Agnes Vanderburg: |
Old film of older woman, Agnes Vanderburg, peeling away bitterroot, looking for heart. |
|
Alyssa's voice over continues: |
Alyssa’s hands working a bitterroot plant, peeling away root to find the heart. She finds it, a small pink seed-like piece, and carefully returns it back into the earth so it will grow again. |
|
Soft instrumental music |
Alyssa, her mother, and a friend walk through the sagebrush |
|
|
The following credits in white text over a black background: |
![]() Principle 4
Principle 4
What You Need to Know About Principle 4: Climate is Variable
This principle relates to some of the differences between weather and climate, how processes like El Nino and the Southern Oscillation influence natural climate variability, and abrupt climate change, which can be triggered by naturally occurring dynamics. Understanding climate variability is critically important in helping scientists tease apart natural variation from human-caused climate change. Click the tabs below to learn more.
- A Little About What Climate Is
Climate is determined by the long-term pattern of temperature and precipitation averages and extremes at a location. Climate descriptions can refer to areas that are local, regional, or global in extent. Climate can be described for different time intervals, such as decades, years, seasons, months, or specific dates of the year. Jump to A Little About What Climate Is
- Climate is NOT Weather
- Natural Climate Variability is NOT Climate Change
Climate change is a significant and persistent change in an area’s average climate conditions or their extremes. Seasonal variations and multi-year cycles (for example, the El Niño) that produce warm, cool, wet, or dry periods across different regions are a natural part of the way climate varies. They do not represent climate change and help scientists tell the difference between climate change that is naturally caused and climate change that is human caused. Jump to Natural Climate Variability is NOT Climate Change
- Global Climate has Changed in the Past and Will Change in the Future
Scientific observations indicate that global climate has changed in the past, is changing now, and will change in the future. The magnitude and direction of this change is not the same at all locations on Earth. Jump to Global Climate has Changed in the Past and Will Change in the Future
- The Earth’s Average Temperature is Warmer Now Than it has Been in 1,300 Years
Based on evidence from tree rings, other natural records, and scientific observations made around the world, Earth’s average temperature is now warmer than it has been for at least the past 1,300 years. Average temperatures have increased markedly in the past 50 years, especially in the North Polar Region. Jump to The Average Temperature is Warmer Now Than it has Been in 1,300 Years
- We have Known for a Long Time How the Greenhouse Effect Works
Natural processes do not explain the rapid climate change observed in recent decades. The only explanation that is consistent with all available evidence is that human impacts are playing an increasing role in climate change. Future changes in climate will be rapid compared to historical changes. Jump to We have Known for a Long Time How the Greenhouse Effect Works
- Carbon Dioxide (CO2) Added to the Atmosphere Stays there for a Century or Longer
Natural processes that remove carbon dioxide from the atmosphere operate slowly when compared to the processes that are now adding it to the atmosphere. Thus, carbon dioxide introduced into the atmosphere today will remain there for a century or more. Other greenhouse gases, including some created by humans, will remain in the atmosphere for thousands of years. Jump to Cabon Dioxide Added to the Atmosphere Stays there for a Century or Longer
Explore this principle by clicking through the bubbles (each takes you to a new concept) at the top of the page.
![]() Principle 4a
Principle 4a
A Little About What Climate Is
Climate is determined by the long-term pattern of temperature and precipitation averages and extremes at a location.
Climate descriptions can refer to areas that are local, regional, or global in extent and can be described for different time intervals, such as decades, years, seasons, months, or specific dates of the year.
But climate is not weather and the two should never be confused. Climate looks at long-term patterns, weather looks at the short term. Read more…
Comparing climates of different planets is useful in developing perspective on climate in general.
For the Earth, one of the most widely used climate classification systems in the world, the Köppen climate classification (see the map below) was developed by Wladimir Köppen around 1900 and is rooted in the concept that native vegetation is the most accurate expression of climate.
Thus, climate zone boundaries are delineated by vegetation distribution, but also take into consideration seasonality and average annual, monthly temperatures and precipitation.
The Köppen system has been updated several times over the years, most recently in 2007.
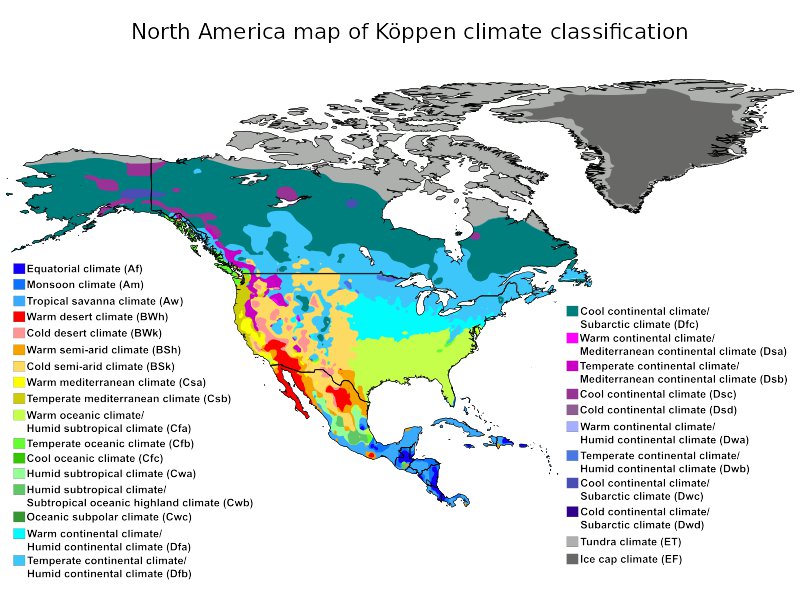
Climates can be classified according to the long-term patterns and the typical ranges of the different climatic variables, such as solar radiation, longwave radiation, air temperature, wind, atmospheric humidity and precipitation. The most widely used applied classification is the Köppen-Geiger system (1936) which groups the climate in 5 main categories: tropical, dry, warm temperate, snow climates and ice climate. These are further subdivided, indicating 12 main different climate zones in the world.
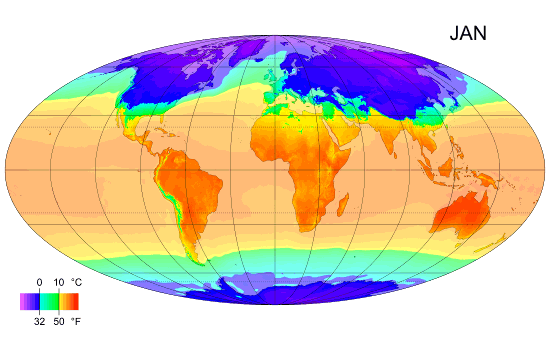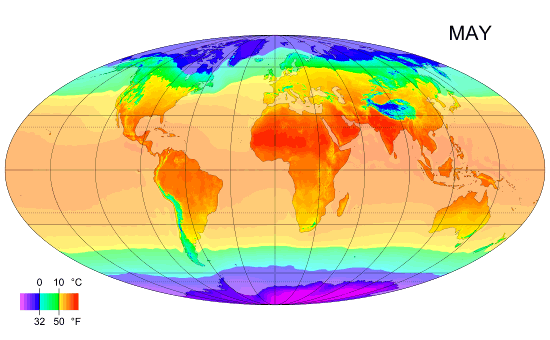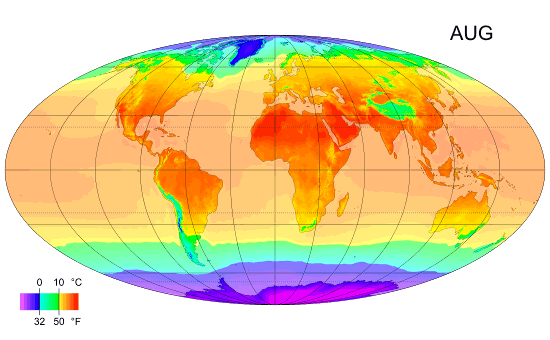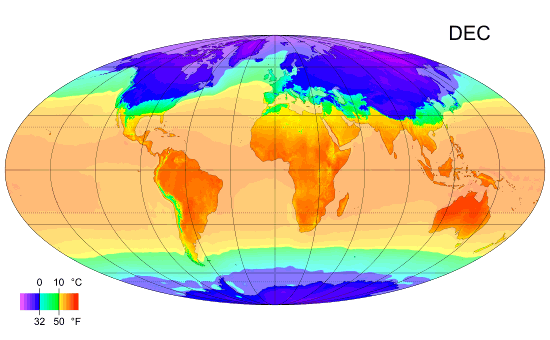
Average monthly temps
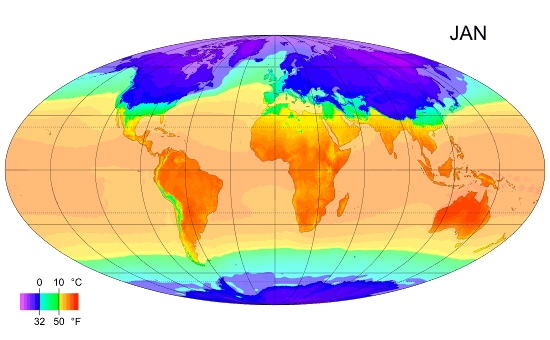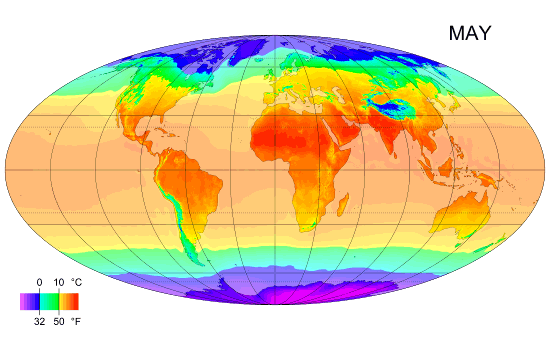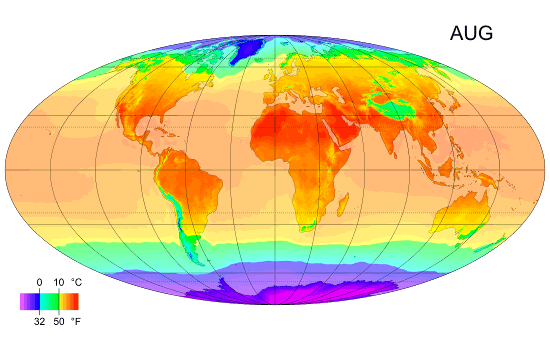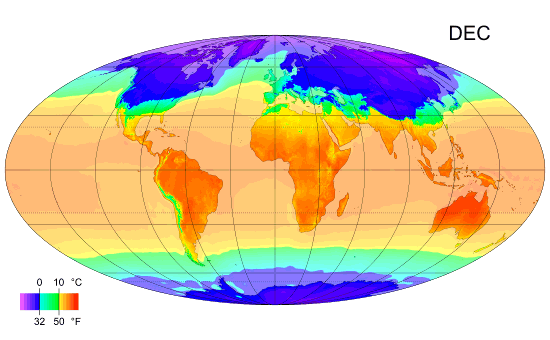
Monthly average surface temperatures from 1961–1990. This is an example of how climate varies with location and season.
History of contributions of planetary studies to the science of climate change
Published: May 13, 2008, 7:48 pm
Updated: July 27, 2012, 8:55 am
Author: Spencer Weart
American Institute of Physics
Venus as imaged by the Magellan mission to Venus. This global view of the surface of Venus is centered at 180 degrees east longitude. (Source: NASA)
A planet is not an object in the laboratory that scientists can subject to different pressures and radiations, comparing how it reacts to this or that. We have only one Earth, and that makes climate science difficult. We can learn much by studying how past climates were different from that of today, and observing how the climate changes in reaction to humanity's "large scale geophysical experiment" of emitting greenhouse gases may teach us a great deal. But these are limited comparisons—different breeds of cat, but still cats. Fortunately, our solar system contains planets with radically different atmospheres.
By the mid 1950s, scientists knew that the atmosphere of Mars was unbreathable—composed mainly of carbon dioxide (CO2), very tenuous and cold, and occasionally stirred up with yellowish dust storms. If Mars had features resembling "canals", as some suspected, they were not full of water for water could not exist as a liquid on the planet's surface. The Mariner 4 spacecraft of 1965, sending back blurry pictures that showed a surface scarred with craters like the Moon, confirmed that the planet was an unlikely abode of life. As for Venus, radio observations published in 1958 showed an amazingly hot climate, with temperatures upwards of 600° Kelvin (K), around the melting point of lead. "It was very disappointing to many people," one of the discoverers recalled, "who were reluctant to give up the idea of a sister planet and perhaps even the possibility of life." Some astronomers worked up arguments that the radio measurements were misleading, representing something in the upper atmosphere, so that life might still exist on Venus. The matter was settled in 1962 when the spacecraft Mariner 2 flew past the planet and detected an unquestionably hot surface.
Already back in 1940, Rupert Wildt had made a rough calculation of the greenhouse effect, caused by the great amount of CO2 others had found in telescope studies of Venus, predicting the effect could raise the surface temperature above the boiling point of water. But raising it as high as 600°K seemed impossible, and nobody mounted a serious attack on the problem; after all, there were very few people in the field of planetary astronomy in those decades. Finally in 1960 a young doctoral student, Carl Sagan, took up the problem and found a solution that made his name known among astronomers. Using what he later recalled as "embarrassingly crude" methods, taking data from tables designed for steam boiler engineering, he confirmed that Venus could indeed be a greenhouse effect furnace. The atmosphere would have to be almost totally opaque, and this "very efficient greenhouse effect" couldn't all be due to CO2—he pointed to absorption by water vapor as the likely culprit.
Dr. Carl Sagan, best known for his acclaimed public television series
Sagan concluded that "Venus is a hot, dry, sandy... and probably lifeless planet." He proposed, most significantly, that the situation was self-perpetuating—the surface of the planet was so hot that whatever water the planet possessed remained in the atmosphere as vapor, helping maintain the extreme greenhouse effect condition. However, it was later found that Sagan was mistaken, for Venus's atmosphere has little water. Today's explanation of the planet's strong greenhouse effect is that Venus has a much denser CO2 atmosphere than assumed by astronomers in the past. But mistakes in science can be as useful as valid results when they stimulate further work and ultimately point in the right direction.
A few researchers tried putting a feedback between temperature and water vapor into a simple system of equations; the results were strange. In 1969, Andrew Ingersoll reported "singularities"—mathematical points where the numbers went out of bounds. This signaled "a profound change in the physical system which the model represents," and Ingersoll pointed to CO2 as the key ingredient in the effect. (The Soviet Venera 4 had penetrated Venus’s atmosphere in 1967 and showed it was mostly CO2, and in 1978 the American Pioneer spacecraft found it was almost entirely CO2.) Sagan had estimated that Venus had started out with roughly the same amount of CO2 as the Earth. On our planet, most of the carbon is locked up in minerals and buried in sediments. The surface of Venus, by contrast, was so hot and dry that carbon-bearing compounds evaporated rather than remaining in the rocks, releasing great amounts of the greenhouse gas into the atmosphere. Perhaps Venus had once enjoyed a climate of the sort hospitable to life, but as water had gradually evaporated into the warming atmosphere, followed by CO2, the planet had fallen into its present hellish state? In a 1971 paper, James Pollack argued that Venus might once have had oceans like Earth's. It seemed that such a "runaway greenhouse" could have turned the Earth too into a furnace, if the starting conditions had been only a little different.
In the late 1970s Michael Hart pursued the idea with a more complex computer model, and concluded that the balance was exceedingly delicate. Hardly any planets in the universe, he said, orbited in the narrow "habitable zone" around a star where life could flourish. For our solar system, the orbits in which a planet would be too close to the Sun—so that at some point the planet would suffer a runaway greenhouse effect from which it could never recover—were separated by only a 5% gap from orbits in which the planet would be so far away that runaway glaciation would freeze any ocean solid. The Earth, then, was a lucky place. However, Hart's calculations were riddled with untested assumptions, and many scientists denied that our situation was so extremely precarious. Nonetheless, Hart defended his ideas energetically among his colleagues, and to the public, including an appearance on television in "Walter Cronkite's Universe." Later calculations disproved Hart's conclusions—a Venus-type runaway on our planet is scarcely possible, even if we burn all available fossil fuels.)
The atmosphere of Venus was filled not only with CO2 but also with an opaque haze. Its nature was unknown, and in the 1960s scientists could only say that the haze was probably caused by some kind of tiny particles. "The clouds on Venus had long been a mystery," as one expert recalled, "in which stratospheric aerosols now appeared to play a key role. The unraveling of the precise role of aerosols in the Venus atmosphere would certainly benefit studies of chemical contamination of Earth's atmosphere." In the early 1970s, ground-based telescope observations produced extraordinarily precise data on the optical properties of these aerosols, and at last they were identified—the haze was made up of sulfur compounds.
The greenhouse effect of the sulfates could be calculated, and by the late 1970s, NASA climate modeler James Hansen stated confidently that the sulfates, together with CO2, "are responsible for the basic climatic state on Venus." (CO2 was by far the largest factor, and the exact effects of sulfates would be debated in many subsequent studies.) Hansen had originally become interested in the greenhouse effect when, in response to Sagan's primitive calculations, he tried to derive a better explanation of why the planet's atmosphere was so hot. Hansen's findings about sulfate aerosols strengthened his belief that these particles could have a significant effect on Earth's climate as well. Sulfates were emitted by volcanoes and, increasingly, by human industry, so perhaps Venus had things to tell us about climate change at home.
The planet Mars as imaged by The Mars Global Surveyor (MGS) Mars Orbiter Camera (MOC) in April, 2003. (Source: NASA)
And then there was Mars—the Red Planet—which also inspired important new thinking about the Earth's atmosphere, even before spacecraft observations. In the 1960s, NASA asked a group of scientists to derive a means of detecting life on Mars. A few noticed that life on Earth makes its presence blatantly evident by driving the atmosphere far from chemical equilibrium. In particular, the abundant oxygen in the air would swiftly drain away, by combining with surface minerals, but the oxygen is renewed by daily emissions from plantlife. Telescopic studies found practically no oxygen in the Red Planet's atmosphere, and, overall, the Martian atmosphere showed no signs of any chemical disequilibrium. Biochemist James Lovelock dismayed his peers by arguing that this showed any search for life on the planet would be fruitless. The sterile atmosphere of Mars, so strikingly different from the Earth's, helped Lovelock, and eventually others, to recognize that life plays a central role in determining the nature of our own planet's atmosphere.
In 1971 the spacecraft Mariner 9, a marvelous jewel of engineering, settled into orbit around Mars and saw... nothing. A great dust storm was shrouding the entire planet. While such storms are rare for Mars, this one was no misfortune, but great luck. Observers immediately saw that the dust had profoundly altered the Martian climate, warming the planet by tens of degrees. The dust settled after a few months, but its lesson was clear—haze could warm an atmosphere. More generally, one studying the climate of any planet would have to take dust very seriously. In particular, it seemed that on Mars the temporary warming had reinforced a pattern of winds, which kept the dust stirred up. It was a striking demonstration that feedbacks in a planet's atmospheric system could flip weather patterns into a drastically different state. This was no longer speculation but an actual event in full view of scientists—"the only global climatic change whose cause is known that man has ever scientifically observed."
Crude speculations about such radical instabilities in the Earth’s own climate had been published independently by two scientists a few years before, about the same time as Ingersoll’s calculation of the Venus runaway greenhouse effect. Pursuing such thoughts before Mariner arrived at Mars, Carl Sagan had made a bold prediction. He suggested that the Red Planet's atmosphere could settle in either of two stable climate states. Besides the current "ice age" there was another possible state, more clement, which might even support life. The prediction seemed to be validated by crisp images of the surface that Mariner beamed home after the dust cleared. The canals some astronomers had imagined were nowhere to be seen, but geologists did see strong signs that vast water floods had ripped the planet in the far past. It was a deadly blow to the old, comforting belief that planets had naturally stable climates, and reinforced the “runaway greenhouse” speculations about Venus that were emerging around the same time.
Calculations by Sagan and his collaborators now suggested that the planet's climate system was balanced so that it could have been flipped from a state with oceans to the present frigid desert, or even back and forth between the two states, by relatively minor changes—changes in its orbit, in the strength of the Sun's radiation, or in the reflection of sunlight off the polar ice cap. These were also, as the authors remarked, "fashionable variables in theories of climatic change on Earth." (Not until 2004 did direct observations on the surface of Mars prove that the planet had indeed once carried standing water, although much farther in the past than Sagan had guessed.)
Speculations published recently about the Earth’s climate suggested possibilities as spectacular as anything proposed for Mars. A small change could start a warming in which the Earth's polar ice caps would shrink, lowering the planet's reflectivity and pushing the warming further into a self-sustaining climate shift. Much the same thing could perhaps happen on Mars, releasing the CO2 frozen at its poles, starting a greenhouse effect process that would melt the ice buried in the soil. In fact, some kind of drastic climate shift had happened on Mars, if evidence of ancient floods is correct. In these arguments, dust stood at the fore, since storms that deposited dust on the polar caps and darkened them seemed the most likely mechanism for pushing the planet into its warm phase.
In later years, spacecraft observed the exotic weather of Venus and Mars in great detail. Meanwhile, as computer models of atmospheres improved, the Earth's two neighbors, plus more exotic planets such as Jupiter, occasionally served as testbeds to probe the limits of the modelers' methods. If a set of equations gave plausible results for such utterly different atmospheres, it created more confidence for their applications on Earth. But the main lesson was a larger one. The idea that feedbacks involving the greenhouse effect could have huge consequences for a planet's climate was no longer mere speculative theory—it was an observation of real events.
Further Reading
![]() Principle 4b
Principle 4b
Climate is NOT Weather
Climate is not the same thing as weather.
Weather is the minute-by-minute variable condition of the atmosphere on a local scale.
Climate is the expected yearly weather conditions established over decades. In other words, if you were planning a trip to a faraway place and had no way of knowing what the weather was going to be like, climate is what you would expect to experience based on long-term weather averages for that place. Climate tells you what clothes to buy. Weather tells you what clothes to wear (that day).
For example, the observed weather in Seattle, Washington, on Saturday, October 16, 2010 was sunny with a high of 47°F. But if you were talking about the climate there, you would say: the average high temperature for Seattle, Washington, on October 16 for the period from 1971 to 2000 is 60°F, a value determined by taking the average of all high temperatures recorded for the thirty October 16ths that have occurred over almost 30 years. Read more…
Even though they are very different, many people share the misconception that weather and climate are basically the same.
This is a big mistake!
While climate and weather are related, there are very different processes at work for the two, and they must be studied and forecasted in different ways.
A couple of key concepts about climate and weather that you should know:
- A spell of unusually cold or warm weather does not prove or disprove human-caused climate change.
Climate is defined as a long-term pattern with naturally occurring variability. For example, a week of very cold, snowy weather, does not say much about the climate. It takes looking at hundreds of weeks of weather, averaging them and determining the long-term trend.- Weather and climate can both vary to a large degree over very small distances. For example, look at the difference between the average winter conditions at these two entrances to Yellowstone National Park, which are just 30 miles apart:
West Yellowstone
Average snow depth: 35 inches
Average temp: -1 to 22 degrees
Gardiner
Average snow depth: 2 inches
Average temp: 14 to 33 degrees - Weather and climate can both vary to a large degree over very small distances. For example, look at the difference between the average winter conditions at these two entrances to Yellowstone National Park, which are just 30 miles apart:
FORECASTING WEATHER AND PREDICTING CLIMATE
Weather forecasters try to answer questions like: What will the temperature be tomorrow? Will it rain? How much rain will we have? Will there be thunderstorms? Today, most weather forecasts are based on models, which incorporate observations of air pressure, temperature, humidity and winds to produce the best estimate of current and future conditions in the atmosphere. A weather forecaster then looks at the model output to figure out the most likely scenario. The accuracy of weather forecasts depend on both the model and on the forecaster's skill. Short-term weather forecasts are accurate for up to a week. Long-term forecasts, for example seasonal forecasts, tend to use statistical relationships between large-scale climate signals such as El Niño and La Niña and precipitation and temperature to predict what the weather will be like in one to six months time.
Climate predictions take a much longer-term view. These predictions try to answer questions like how much warmer will the Earth be 50 to 100 years from now? How much more precipitation will there be? How much will sea level rise? Climate predictions are made using global climate models. Unlike weather forecast models, climate models cannot use observations because there are no observations in the future.
NASA - What's the Difference Between Weather and Climate?
Soource: https://www.nasa.gov/mission_pages/noaa-n/climate/climate_weather.htm
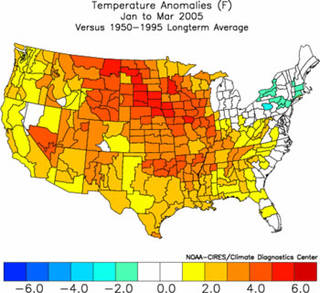
Latest three month average temperature and precipitation anomalies for the United States.
Credits: NOAA
The difference between weather and climate is a measure of time. Weather is what conditions of the atmosphere are over a short period of time, and climate is how the atmosphere "behaves" over relatively long periods of time.
When we talk about climate change, we talk about changes in long-term averages of daily weather. Today, children always hear stories from their parents and grandparents about how snow was always piled up to their waists as they trudged off to school. Children today in most areas of the country haven't experienced those kinds of dreadful snow-packed winters, except for the Northeastern U.S. in January 2005. The change in recent winter snows indicate that the climate has changed since their parents were young.
If summers seem hotter lately, then the recent climate may have changed. In various parts of the world, some people have even noticed that springtime comes earlier now than it did 30 years ago. An earlier springtime is indicative of a possible change in the climate.
In addition to long-term climate change, there are shorter term climate variations. This so-called climate variability can be represented by periodic or intermittent changes related to El Niño, La Niña, volcanic eruptions, or other changes in the Earth system.
What Weather Means
Weather is basically the way the atmosphere is behaving, mainly with respect to its effects upon life and human activities. The difference between weather and climate is that weather consists of the short-term (minutes to months) changes in the atmosphere. Most people think of weather in terms of temperature, humidity, precipitation, cloudiness, brightness, visibility, wind, and atmospheric pressure, as in high and low pressure.
In most places, weather can change from minute-to-minute, hour-to-hour, day-to-day, and season-to-season. Climate, however, is the average of weather over time and space. An easy way to remember the difference is that climate is what you expect, like a very hot summer, and weather is what you get, like a hot day with pop-up thunderstorms.
Things That Make Up Our Weather
There are really a lot of components to weather. Weather includes sunshine, rain, cloud cover, winds, hail, snow, sleet, freezing rain, flooding, blizzards, ice storms, thunderstorms, steady rains from a cold front or warm front, excessive heat, heat waves and more.
In order to help people be prepared to face all of these, the National Oceanic and Atmospheric Administration's (NOAA) National Weather Service (NWS), the lead forecasting outlet for the nation's weather, has over 25 different types of warnings, statements or watches that they issue. Some of the reports NWS issues are: Flash Flood Watches and Warnings, Severe Thunderstorm Watches and Warnings, Blizzard Warnings, Snow Advisories, Winter Storm Watches and Warnings, Dense Fog Advisory, Fire Weather Watch, Tornado Watches and Warnings, Hurricane Watches and Warnings. They also provide Special Weather Statements and Short and Long Term Forecasts.
NWS also issues a lot of notices concerning marine weather for boaters and others who dwell or are staying near shorelines. They include: Coastal Flood Watches and Warnings, Flood Watches and Warnings, High Wind Warnings, Wind Advisories, Gale Warnings, High Surf Advisories, Heavy Freezing Spray Warnings, Small Craft Advisories, Marine Weather Statements, Freezing Fog Advisories, Coastal Flood Watches, Flood Statements, Coastal Flood Statement.
Who is the National Weather Service?
According to their mission statement, "The National Weather Service provides weather, hydrologic, and climate forecasts and warnings for the United States, its territories, adjacent waters and ocean areas, for the protection of life and property and the enhancement of the national economy. NWS data and products form a national information database and infrastructure which can be used by other governmental agencies, the private sector, the public, and the global community."
To do their job, the NWS uses radar on the ground and images from orbiting satellites with a continual eye on Earth. They use reports from a large national network of weather reporting stations, and they launch balloons in the air to measure air temperature, air pressure, wind, and humidity. They put all this data into various computer models to give them weather forecasts. NWS also broadcasts all of their weather reports on special NOAA weather radio, and posts them immediately on their Interactive Weather Information Network website at: http://iwin.nws.noaa.gov/iwin/graphicsversion/bigmain.html.
What Climate Means
In short, climate is the description of the long-term pattern of weather in a particular area.
Some scientists define climate as the average weather for a particular region and time period, usually taken over 30-years. It's really an average pattern of weather for a particular region.
When scientists talk about climate, they're looking at averages of precipitation, temperature, humidity, sunshine, wind velocity, phenomena such as fog, frost, and hail storms, and other measures of the weather that occur over a long period in a particular place.
For example, after looking at rain gauge data, lake and reservoir levels, and satellite data, scientists can tell if during a summer, an area was drier than average. If it continues to be drier than normal over the course of many summers, than it would likely indicate a change in the climate.
Why Study Climate?
The reason studying climate and a changing climate is important, is that will affect people around the world. Rising global temperatures are expected to raise sea levels, and change precipitation and other local climate conditions. Changing regional climate could alter forests, crop yields, and water supplies. It could also affect human health, animals, and many types of ecosystems. Deserts may expand into existing rangelands, and features of some of our National Parks and National Forests may be permanently altered.
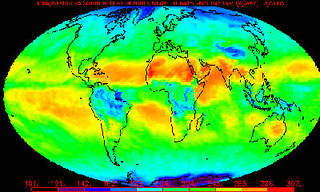
An example of a Monthly Mean Outgoing Longwave Radiation (OLR) product produced from NOAA polar-orbiter satellite data, which is frequently used to study global climate change.
Credits: NOAA
The National Academy of Sciences, a lead scientific body in the U.S., determined that the Earth's surface temperature has risen by about 1 degree Fahrenheit in the past century, with accelerated warming during the past two decades. There is new and stronger evidence that most of the warming over the last 50 years is attributable to human activities. Yet, there is still some debate about the role of natural cycles and processes.
Human activities have altered the chemical composition of the atmosphere through the buildup of greenhouse gases – primarily carbon dioxide, methane, and nitrous oxide. The heat-trapping property of these gases is undisputed although uncertanties exist about exactly how Earth's climate responds to them. According to the U.S. Climate Change Science Program (http://www.climatescience.gov), factors such as aerosols, land use change and others may play important roles in climate change, but their influence is highly uncertain at the present time.
Who Studies Climate Change?
Modern climate prediction started back in the late 1700s with Thomas Jefferson and continues to be studied around the world today.
At the national level, the U.S. Global Change Research Program coordinates the world's most extensive research effort on climate change. In addition, NASA, NOAA, the U.S. Environmental Protection Agency (EPA) and other federal agencies are actively engaging the private sector, states, and localities in partnerships based on a win-win philosophy and aimed at addressing the challenge of global warming while, at the same time, strengthening the economy. Many university and private scientists also study climate change.
What is the U.S. Global Change Research Program?
The United States Global Change Research Program (USGCRP) was created in 1989 as a high-priority national research program to address key uncertainties about changes in the Earth's global environmental system, both natural and human-induced; to monitor, understand, and predict global change; and to provide a sound scientific basis for national and international decision-making.
Since its inception, the USGCRP has strengthened research on global environmental change and fostered insight into the processes and interactions of the Earth system, including the atmosphere, oceans, land, frozen regions, plants and animals, and human societies. The USGCRP was codified by Congress in the Global Change Research Act of 1990. The basic rationale for establishing the program was that the issues of global change are so complex and wide-ranging that they extend beyond the mission, resources, and expertise of any single agency, requiring instead the integrated efforts of several agencies.
Some Federal Agencies Studying Climate
In the 1980s the National Weather Service established the Climate Prediction Center (CPC), known at the time as the Climate Analysis Center (CAC). The CPC is best known for its United States climate forecasts based on El Niño and La Niña conditions in the tropical Pacific.
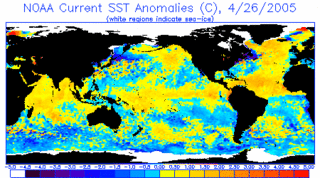
Image Above: The operational SST anomaly charts are useful in assessing ENSO (El Niño - Southern Oscillation) development, monitoring hurricane "wake" cooling, and even major shifts in coastal upwelling.
Credits: NOAA
CPC was established to give short-term climate prediction a home in NOAA. CPC's products are operational predictions or forecasts of how climate may change and includes real-time monitoring of climate. They cover the land, the ocean, and the atmosphere, extending into the upper atmosphere (stratosphere). Climate prediction is very useful in various industries, including agriculture, energy, transportation, water resources, and health.
NASA has been using satellites to study Earth's changing climate. Thanks to satellite and computer model technology, NASA has been able to calculate actual surface temperatures around the world and measure how they've been warming. To accomplish the calculations, the satellites actually measure the Sun's radiation reflected and absorbed by the land and oceans.NASA satellites keep eyes on the ozone hole, El Nino's warm waters in the eastern Pacific, volcanoes, melting ice sheets and glaciers, changes in global wind and pressure systems and much more.
At the global level, countries around the world have expressed a firm commitment to strengthening international responses to the risks of climate change. The U.S. is working to strengthen international action and broaden participation under the support of the United Nations Framework Convention on Climate Change.
Today, scientists around the world continue to try and solve the puzzle of climate change by working with satellites, other tools and computer models that simulate and predict the Earth's conditions.
For information about the U.S. Global Change Research Program, please visit:
http://www.usgcrp.gov/
For information about NASA's study of Earth's climate, please visit on the Internet:
http://www.nasa.gov/vision/earth/features/index.html
For a review of 2004's Global Temperature, please visit:
http://www.nasa.gov/vision/earth/lookingatearth/earth_warm.html
For information about NASA, please visit on the Internet:
http://www.nasa.gov
For information about the National Weather Service, please visit on the Internet:
http://www.nws.noaa.gov/
For immediate watches and warnings, visit the NWS Interactive Weather Information Network website at:
http://iwin.nws.noaa.gov/iwin/graphicsversion/bigmain.html
To find a NOAA weather radio station near you:
http://www.nws.noaa.gov/nwr/
For a glossary of weather terms, please visit the National Weather Service Weather Glossary on the Internet at:
http://www.nws.noaa.gov/glossary/
Rob Gutro
NASA's Earth-Sun Science News Team/SSAI
NASA Goddard Space Flight Center, Greenbelt, Md., and excerpts from NOAA's CPC web page, and the U.S. EPA web page. 2/2005
Edits: Dr. J. Marshall Shepherd, NASA/GSFC, Drew Shindell, NASA/GISS, Cynthia M. O'Carroll, NASA/GSFC
Last Updated: May 13, 2015
Editor: NASA Administrator
![]() Principle 4c
Principle 4c
Natural Climate Variability is NOT Climate Change
When scientists talk about climate change, they are talking about a long-term change in an area’s average climate conditions. They are not talking about things like the differences between seasons or cycles like El Niño.
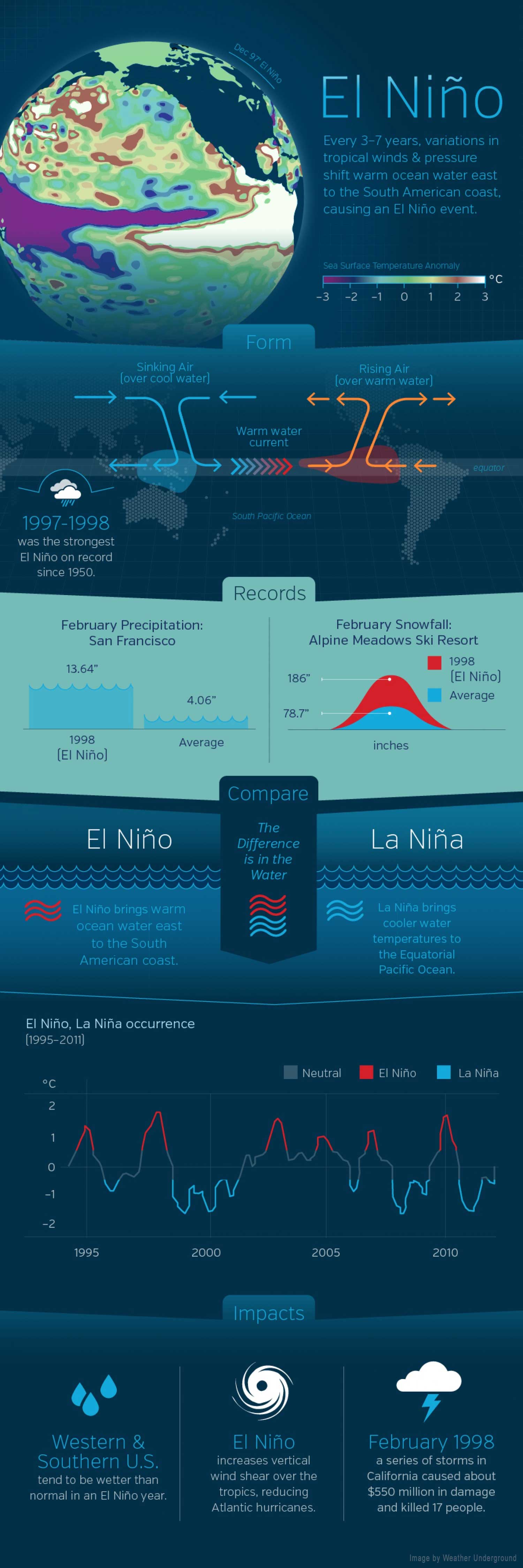
Multi-year, periodic cycles like El Niño produce warm, cool, wet, or dry periods across different regions over one or more years. These are a natural part of the way climate varies and help scientists tell the difference between climate change that is naturally caused and climate change that is human caused, but they do not represent climate change. Read more…
On land, especially at high latitudes and elevations, seasonal changes that occur during the normal course of the year dance are somewhat predictable. Precipitation and temperature patterns occur at more or less the same time each year (for example, winter and summer) with organisms and the land itself responding to the seasonal fluctuations.
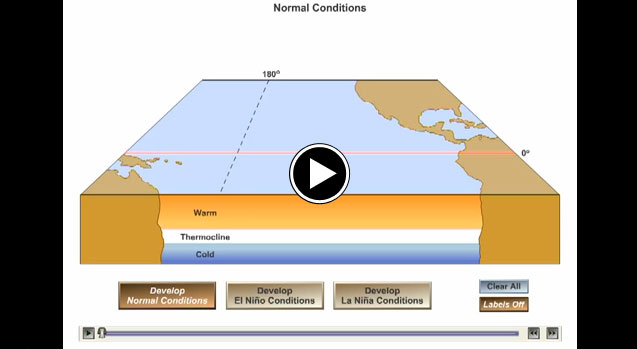
The ocean retains and releases heat differently than land. While the annual cycle is important throughout the ocean, its surface waters are separated from colder, deeper water by a thermocline. The upwelling of cold waters, especially along the equator in the eastern half of the Pacific basin, can be blocked when sea level is high, resulting in warm events like El Niño. The two videos and the link to the poster below illustrate and explain these multi-year El Niño cycles. These cycles are not examples of climate change.
What to expect when you’re expecting El Niño
- By Ben Goldfarb
- Source: https://www.hcn.org/blogs/goat/what-to-expect-when-youre-expecting-el-nino-1
The data are trickling in, and with each passing day it seems more certain: 2014 is going to be an El Niño year, and probably a big one. What does that mean for your Western state?
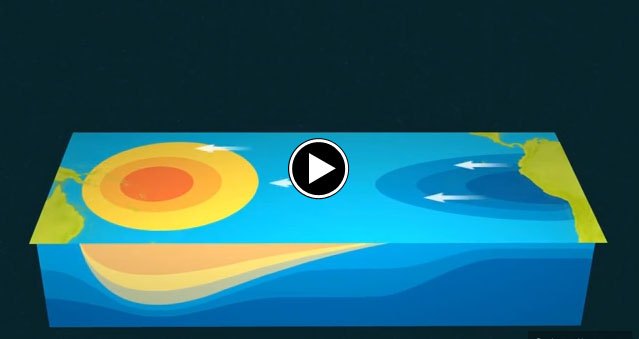
First, a quick primer on the science behind The Niño. In normal years, prevailing winds in the Pacific Ocean push warm water to the west, toward Indonesia, leaving space for deeper cold water to rise off the coast of South America. When those trade winds slacken or about-face – a phenomenon that appears to be occurring right now – fasten your seat belts and prepare for a bumpy El Niño ride. The reversed winds drive warm waters eastward and up toward the ocean’s surface, where they come into contact with the atmosphere, increase air temperatures, and send global weather patterns haywire, generating storms, droughts, and heat waves. Voila – you’re a meteorologist!
“I think there’s no doubt that there’s an El Niño underway,” climate scientist Kevin Trenberth of the U.S. National Center for Atmospheric Research recently told Wired. “The question is whether it’ll be a small or big one.” What makes scientists like Trenberth so confident? The Pacific is already heating up. In the below map, yellow and orange areas represent temperature anomalies – places where the surface of the ocean is unusually warm for this time of year. You're about to see a whole lot of yellow:

While it’s still a little early to be making definitive predictions about the magnitude of the coming El Niño, it may turn out to be a beast to rival the 1997-98 event, which cost the U.S. an estimated $10 billion in storm damages and crop losses. (Although El Niño typically transpires every two to seven years, climate change could be increasing the frequency of super-strong events.) As Eric Holthaus explained in Slate, the subsurface blob of warm water now drifting toward the Americas is enormous – large enough to cover the entire United States to a depth of 300 feet. That immense anomaly could make 2014 the hottest year in recorded global history, and 2015 might be even steamier.
Every El Niño is different, of course, and extrapolating from previous events involves a whole lot of semi-educated guesswork. As some sage once said, “It’s hard to make predictions, especially about the future.” With that caveat out of the way, let’s comb through the historic record to understand what havoc the coming disturbance might wreak upon the American West.

During an El Niño year, the jetstream ducks further south than usual, conveying heavy rains to California. Image courtesy of NOAA.
Oregon: Oh look, another northwestern state that’s in for lackluster snowpack. El Niño years tend to produce lower stream flows in the Columbia Basin, spelling trouble for dam operators and fishermen alike. Gulp.
Idaho: Brace yourself, Idahoans: you’re in for a dry winter. Hey, don’t shoot the messenger – check out the below map. The same meandering jetstream that brings rain to California tends to shift wet weather away from the Pacific Northwest, Idaho included. Plenty of precipitation fell on the Gem State this winter, so hopefully the reservoirs contain a bit of a buffer.

Green areas receive more rain during El Niño years; brown areas receive less. Right under the title of this image are a group of years; each of those years was an El Nino year. Image courtesy of NOAA.

Green bars represent unusually wet winters during El Niño years; brown bars represent unusually dry winters. Each set of bars represents a different region of New Mexico. Wet winters are clearly more common than dry throughout the state. Image courtesy of NOAA.
Washington: Inflicting floods and drought is one thing, El Niño, but when your warm waters start messing with salmon, you’ve gone too far. Previous El Niños have been blamed for increased mortality and decreased growth in Pacific salmon; in 1994, the salmon situation became so dire that the federal government declared it a disaster. Columbia Basin returns have spiked in recent years, partly thanks to favorable (i.e., cool) ocean conditions, but don’t bank on another epic run of fall Chinook in 2015.
California: If there’s one faction sure to welcome El Niño with open arms, it’s California farmers, who have spent the last year locked in the mother of all droughts. El Niño should provide some welcome relief: During past events, the jetstream has migrated south into California, bearing ample rain. In fact, Cali could go straight from drought to flood, as torrential storms in 1998 destroyed homes and forced the evacuation of towns. At least sport fishermen will be satisfied: the warming Pacific conveys exotic gamefish to the California coast.
So there you have ‘em – your fearless, west-wide El Niño predictions. And if our forecasts don’t jibe with your memories of El Niños past, let us know in the comments.
Ben Goldfarb is an editorial intern at High Country News. He tweets @bengoldfarb13.
El Niño conditions are growing stronger
By Mike Carlowicz,
NASA’s Earth Observatory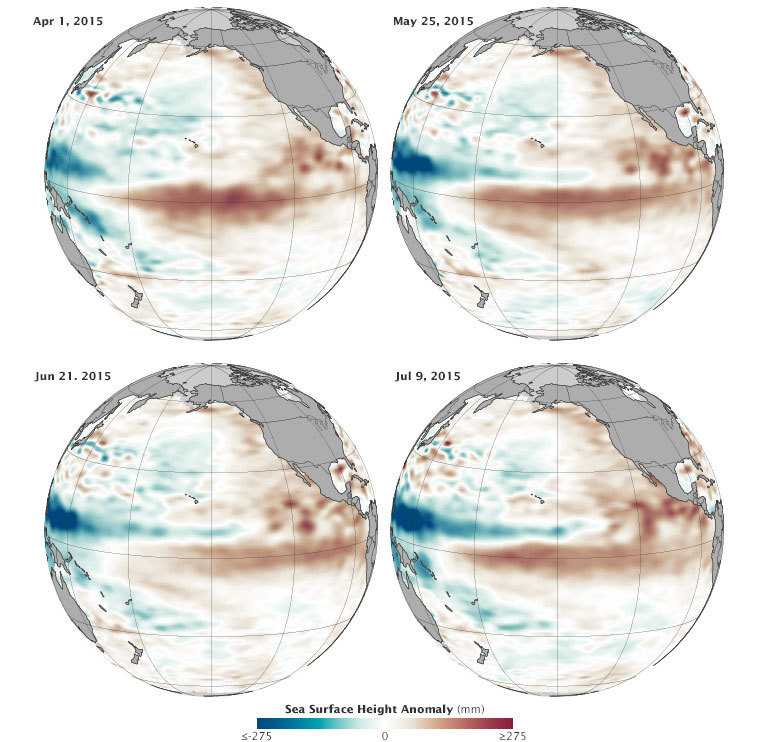
NASA Earth Observatory image by Jesse Allen, using Jason-2 data provided by Akiko Kayashi and Bill Patzert, NASA/JPL Ocean Surface Topography Team. Credit: NASA. Data acquired April to July 2015.
When scientists declared in March 2015 that El Niño conditions had developed in the Pacific Ocean, the consensus was that the event was too weak and too late to have much effect on North America. But in the past several months, warm water has been sloshing from the western Pacific toward the Americas and El Niño has strengthened. Surface waters have grown significantly warmer in the central and eastern Pacific, and conditions have become somewhat cooler and drier in the west. By the end of July 2015, scientists at NASA and other agencies started to see some similarities between current conditions and the development of the potent El Niño event of 1997–98.
“We have not seen a signal like this in the tropical Pacific since 1997,” said Bill Patzert, a climatologist at NASA’s Jet Propulsion Laboratory. “It’s no sure bet that we will have a strong El Niño, but the signal is getting stronger. What happens in August through October should make or break this event.”
The maps on this page provide NASA’s view of Pacific Ocean conditions. At the top of the page, you can see snapshots of the averaged sea surface height anomalies at four different moments since March 2015. Shades of red indicate where the ocean stood above normal sea level because warmer water expands to fill more volume (thermal expansion). Shades of blue show where sea level and temperatures were lower than average (thermal contraction). Normal sea-level conditions appear in white. The maps are based on altimetry measurements made by the OSTM/Jason-2 satellite and analyzed by scientists at NASA’s Jet Propulsion Laboratory.
Below you can see an animation of the same data from January 1 through July 31, 2015. Note how pulses of warmer water seem to move from west to east across the Pacific basin. There is a subtle signal in January, and then increasingly stronger pulses in March, May, and July.
The pulses of warmer water moving across the ocean are Kelvin waves. Sea level is naturally higher in the western Pacific; in fact, it is roughly 40 to 50 centimeters (15-20 inches) higher near Indonesia than off of Ecuador. Much of this difference is due to tropical trade winds, which predominantly blow from east to west across the Pacific Ocean, piling up the water near Asia and Oceania. When those trade winds ease and bursts of wind come out of the west, warm water from the western Pacific sloshes east in vast and deep waves and evens out sea level a bit. As the warm water piles up in the east, it suppresses the natural upwelling that usually keeps waters cooler along the Pacific coasts of the Americas.
The seas and skies have been doing just that in 2015. According to observations compiled by the National Oceanic and Atmospheric Administration (NOAA), at least three sizable west wind bursts have occurred in the Pacific since March, and each came shortly before a Kelvin wave rolled across the basin. Those waves have raised water temperatures in the central and eastern Pacific by as much as 1.6 degrees Celsius (2.9° Fahrenheit) above normal, making for a “strong” El Niño. The warmers waters off the west coast of the Americas have led to warmer and more humid weather ashore, as well as soaking bursts of rain. At the same time, Indonesia and other parts of the western Pacific have been unusually dry.
“Sea surface temperatures in the eastern Pacific have been waxing and waning,” Patzert said. “Right now they are waxing.” But will sea surface conditions and winds amplify the warming signal and produce an El Niño to rival 1997–98?
Below you will find a comparison of sea surface height in the Pacific as measured at the end of July in 1997 and 2015. The left-side measurements come from the TOPEX/Poseidon mission, while the right side is from Jason 2. Comparing the two years, 1997 seems slightly less intense. But trade winds collapsed and the eastern Pacific warmed dramatically from August through November 1997, setting the stage for a turbulent winter that brought flooding rains and landslides across the West Coast of North and South America. (Click here to see an animation of 1997 conditions compared to 2015.)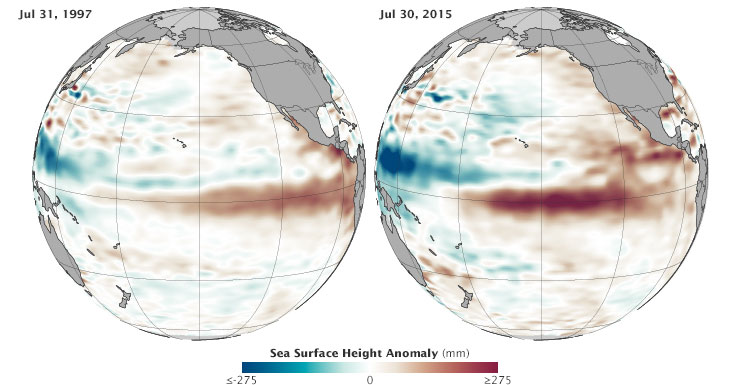
NASA Earth Observatory image by Jesse Allen, using Jason-2 data provided by Akiko Kayashi and Bill Patzert, NASA/JPL Ocean Surface Topography Team. Data acquired January to July 2015.
“This El Niño is getting billed as the ‘great wet hope.’ Many people in the American West are looking to El Niño to save them from drought,” Patzert noted. But he cautioned that “1997 was mayhem,” and drenching rains on a parched landscape are just as hazardous as a drought.
El Niño typically peaks between December and April, so only time will tell if this event will be potent. Many of the models and observations suggest it will be, but other factors such as the “warm blob” in the North Pacific and the apparent shift in the Pacific Decadal Oscillation could affect further development.
“With hopes for drought relief running so high in California, it can’t be stressed enough that El Niño shifts the odds but doesn’t guarantee the roll of the meteorological dice in any particular winter,” wrote meteorologist and blogger Bob Henson.
Scientists from NASA’s Goddard Space Flight Center and other institutions recently found that California has accumulated a “rain debt” of about 50 centimeters (20 inches) between 2012 and 2015. That is the amount that would normally fall in an entire year in the state.
And even if the rains come, they do not necessarily solve drought problems in areas that rely on snow pack for summer supplies. “It took a long time to get into this drought,” Patzert added, “and it is more of a systemic problem than just a lack of rain or snowfall.”
References and related reading
- NASA JPL Ocean Surface Topography from Space (2015) El Niño 2015. Accessed August 3, 2015.
- NOAA Climate.gov (2015, July 7) Keep calm and stop obsessing over weekly changes in ENSO. Accessed August 3, 2015.
- NOAA Climate.gov (2015, July 9) July 2015 El Niño Update: Bruce Lee? Accessed August 3, 2015.
- NASA Earth Observatory (2015, April 18) Warm Water and Strange Weather May Be Connected.
- NASA Earth Observatory (2015, March 25) Weak El Niño, but Hints of Pacific Change.
- NASA Earth Observatory (2009) World of Change: El Niño, La Niña, and Rainfall.
- NASA Precipitation Measurement Missions (2015) NASA Calculates California’s Rain Debt. Accessed August 3, 2015.
- Los Angeles Times (2015, July 29) El Niño contributing to ‘monsoon on steroids’ behind Southland’s humid weather. Accessed August 3, 2015.
- Mother Jones (2015, July 30) El Niño vs. the Blob: Here’s Why California’s Drought Probably Won't End Anytime Soon. Accessed August 3, 2015.
- WunderBlog, via Weather Underground (2015, July 28) What to Expect from El Niño: North America. Accessed August 3, 2015.
A thermocline is a thin but distinct layer in an ocean in which temperature changes more rapidly with depth than it does in the layers above or below. In the ocean, the thermocline can be thought of as an invisible blanket which separates the upper mixed layer from the calm deep water below. 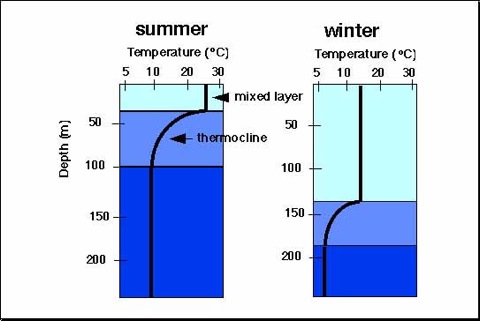
![]() Principle 4d
Principle 4d
Global Climate has Changed in the Past and Will Change in the Future
Scientific observations show that the earth’s climate has changed in the past, is changing now, and will change in the future.
But the magnitude and direction of that change varies depending on where you are on the planet.
So just as climates across the Earth vary widely, the changes that accompany recent climate change are also varied. Read more…
While globally there is a major warming trend, some regions are getting wetter and cooler, while others are getting warmer and drier.
It was once thought that climate was generally steady, even-keeled, but we now know that climate change can occur abruptly, as it has many times in the Earth’s past.
As the National Oceanic and Atmospheric Administration (NOAA) states: Over the course of Earth's 4.55 billion orbits around the sun, there were periods when major continental ice sheets were dominant and periods when temperatures were higher and so were sea levels.
Some researchers theorize that during a prolonged cold period between 850-550 million years ago, the world was dominated by ice. This has been called the “Snowball Earth Hypothesis”.
Other researchers claim that the geologic record does not support the theory of a prolonged cold period of 300 million years, but rather was between two and four periods of glaciation with sustained "interglacial" warm periods lasting tens of millions of years.
Click on a topic to learn more about the different cycles of both weather and climate.
Driven by the Earth's spin on its axis, the diurnal cycle of night and day is a powerful driver of variability of weather. A day is actually 0.0027 of a year, more than two orders of magnitude less than a year. In general, the daily weather cycle goes like this:
Sunrise
Sunlight hits the Earth’s surface.
Mid morning
Temperature rises most quickly during this time.
High noon
Hearted air circulates in the bottom 1 km of the atmosphere.
Afternoon
Temperatures are hottest during mid afternoon.
Sunset
At sunset, sunlight stops hitting the earth. Temperatures fall most quickly around sunset as the Earth’s surface and atmosphere radiate heat.
Midnight
At midnight, the lowest level of the atmosphere becomes cooler. In the middle of the night, the Earth, water surfaces and human communities release store heat.
Dawn
Right before dawn, temperatures are coldest around dawn
The annual revolution of the Earth around the sun and seasonal cycles of atmosphere and oceans dominate annual climate and weather dynamics. Volcanic eruptions can also play a role in variability because they emit aerosols.
Solstice Dec. 21 or 22
The winter solstice is the shortest day in Northern Hemisphere, the longest day in the Southern. The northern Hemisphere tilts away from sun, receiving less direct solar radiation, while the Southern Hemisphere tilts towards the sun. Blizzards and ice storms can occur throughout the boreal (Northern Hemisphere) winter months in higher latitudes and elevations.
Spring Equinox March 20 or 21
When the sun's direct rays pass the equator, the length of day and night are the same. (Equinox means " equal night"). Without sufficient spring precipitation, droughts may occur later in the year as evapotranspiration increases. In regions where winter and spring precipitation falls as snow, runoff peaks start in the Spring in the southern regions and progresses northward and upward in elevation as the season progresses.
Sostice June 21 or 22
The summer solstice is the longest day in Northern Hemisphere, the shortest day in the Southern. Northern Hemisphere tilts toward the sun, receiving more intense direct solar radiation, while the Southern Hemisphere tilts away from the sun and experiences winter. In regions where winter and spring precipitation falls as snow, there is often a peak in snow melt runoff around the solstice. Droughts, flash floods, forest fires and hurricanes are all climate-related events that usually occur during the summer months and into the fall.
Wet season during the Indian Monsoon generally begins in June and goes through September.
Autumn Equinox Sept 22 or 23
Sun's direct rays pass the equator and length of day and night are the same. The seasonal cooling of the climate during autumn may include the end of the growing season in some climates. Hurricane activity may extend well into the Autumn season.
The oceans, with their great heat capacity and slowly changing properties, give rise to cycles in climate like El Nino and La Nina. Abrupt climate changes can occur within a ten-year period.
2001
Severe flooding impacts hundreds of thousands in Mozambique and other southeastern African nations in February-April.
Active hurricane season with 15 storms in Atlantic.
Tropical Storm Allison drenches Louisiana and southeast Texas; $5 billion dollars in damages and 41 lives lost.
Southeast Asian drought centered in Afganistan that began in 1998 continues.
Millions in Vietnam and Cambodia effected by flooding in August-October.
2000
Wildfires in the Western US burn over 6 million acres and cost over $1 billion to fight.
Severe drought affects parts of US and Asia, while flooding occurs in SW Asia.
Image of wildfire aftermath from FEMA.
1999
La Niña linked to droughts conditions in much of US and higher than normal precipitation in Pacific Northwest.
Severe windstorms uproot trees in France in December.
Global mean temperature (Land and Ocean combined) for year is 5th
warmest on record since 1880 and second warmest for US, with 1998 being the warmest.
1998
January: Severe Ice Storm hits NE US and Canada, leaving millions without electricity, triggering flooding.
November: Hurricane Mitch devastates Central America, killing estimated 11,000, the greatest loss of life in the western hemisphere from a tropical system since 1780.
1997
Impact of 1997-1998 ENSO warm event estimated at $25 to $33 billion.
Flooding in California and Dakotas cost $6.7 billion.
1996
Hurricanes Bertha and Fran hit the North Carolina resulting in major flooding. Total damages in the United States over $3.5 billion.
1995
Atlantic Hurricane Season second busiest hurricane season since 1871. Nineteen named storms, with 11 of which reached hurricane strength causing $8 billion in damage.
July: Heat wave hits central US, causing 830 deaths, 525 of them in Chicago.
December- Intense storm with heavy rains strikes Pacific Northwest, causing 6 deaths.
1994
March: Severe ice storm in sout heast US causes an estimated $3 billion in damages.
1993
March: "Storm of the Century" hits US east coast with 270 missing, including 14 in Florida who die in tornadoes.
Summer: U. S. Midwest Flood of 1993 reported 48 deaths and $18 billion in damages.
1992
August: Hurricane Andrew hits Florida, killing 54, costing $25 billion. Had the eye of the storm been a few miles further north, downtown Miami might have been hit and the death toll and destruction far higher.
Sept.: Hurricane Iniki affects three Hawaiian Islands and results in over $1 billion in damage.
1991
April: Bangladesh Cyclone kills over 138,000.
October: Typhoon Thelma, hits Philippines and 6000 people die.
October: Oakland Firestorm kills 25 and costs $2.5 billion
November: "Halloween Nor'Easter" later called the "Perfect Storm" develops off Nova Scotia.
Longer than most human life-spans, the period of a century offers a wider perspective of oceanic and atmospheric patterns that influence climatic variability than the scope of a decade allows. Patterns that occur between the decadal and centennial (10-100 year) scales include Pacific Decadal Oscillation (PDO) and North Atlantic Oscillation (NAO).
2000s
$48 billion in losses due to drought
$18 billion in losses due to flooding
$52 billion in losses from severe storms
$328 billion in losses from tropical cyclones
$15 billion in losses from wildfires
1990s
Globally averaged as the warmest decade in the past 140 years, with 1998 being the warmest year on record.
1992- Hurricane Andrew causes $25 billion in damages.
1993- Great Midwest Flood along the Mississippi and its tributaries caused $18B in damages. Considered to be greater than a 500 year flood event.
1997-98- El Niño causes $25-33 billion in damages
1980s
Globally averaged second warmest decade in the past 140 years.
1982-1983 El Niño severely impacts fishing industry in Ecuador and Peru, disrupting weather throughout the Pacific Basin. Estimated $21 billion in damages in 1997.
1970s
Beginning ~30 year positivie trend in the NAO/AO (North Atlantic Oscillation/Arctic Oscillation) that strongly contributed to winter/spring warming over northwestern Europe.
1971- Flooding in Vietnam kills 100,000.
1972- Blizzard in Iran leaves 4000 dead.
1977- The "North Pacific" climatic regime shift making the end of a ~30 year cool phase of the PDO (Pacific Decadal Oscillation) and the beginning of ~21 year warm phase of the PDO; extensive drought conditions in the western U.S.
1978- New England Blizzard dumps up to 38 inches of snow in Boston area.
1960s
1969- Hurricane Camille slams into Mississippi at nearly 200 mph with storm tides of 25 feet. Heavy rains and flooding followed, with 28 inches of rain in central Virginia.
1950s
Nov. 1950- Severe storm event produces blizzard and severe winds in 22 states.
Dec. 1952- Great Smog of London directly kills 4000, with 4000 additional deaths from related causes. Also see NPR story on the Killer Fog of '52.
Jan.-Feb. 1953- Storm surges in Europe cause nearly 2000 deaths.
Sept. 1958- Typhoon Vera hits Japan, killing nearly 5,000, leaving 1.5 million homeless.
Droughts in mid-1950s in western US motivate intense period of dam-building for water storage and delivery.
1940s
Global cooling occurs between mid-1940s and early 1970s.
1941-1942- Chinese Drought causes 3 million to perish due to starvation.
1930s
1930s- Dust Bowl drought impacts 100 million acres of Great Plains.
1931- Flooding along Yangtze River, impacting millions of Chinese.
1935- Florida Keys Hurricane first of two Category 5 hurricanes to make landfall in U.S. during 20th Century.
1938- New England Hurricane rips through southern New England.
1920s
1922- Colorado River Compact signed allocating water of the Colorado River Basin to states in the basin and Mexico; estimates of flows based on data from the wet period prior to the signing of the compact.
1925- Tri-State Tornado claims 695 lives during 3.5 hours, leaving 219-long track through Ohio Valley.
1928- Great Okeechobee Flood and Hurricane kills up to 3,500.
1910s
1917-1920- First case of Spanish Influenza pandemic occurs during winter flu season in March of 1918 when the first case was reported at Fort Riley, Kansas. By October, 195,000 dead in U.S. and by 1920, some 20 million dead worldwide. (Gross-Schulman, 1998 )
1900s
Sept. 1900- Hurricane hits Galveston, Texas with surge wave and resulting floods killing over 8000. (see "Special Report on the Galveston Hurricane of September 8, 1900" by Isaac M. Cline from NOAA).
1907- Famine in China caused 24 million to die of starvation.
In the past 1000 revolutions around the sun (one thousand years), the Earth has experienced a wide variety of human activity against a background of climate change and variability. Drought—some lasting several decades—impacted cultures such as the Ancient Pueblo peoples who inhabited parts of the American Southwest a millennium ago.
Paleoclimatologists use clues from proxy records such as tree rings and layers of sediment and ice to piece together past climate patterns and events over the course of a millennium, far beyond the limited records from modern instruments such as rain gauges and thermometers.
1900 (20th Century)
Severe weather and climate events have increasing impact on society and environment as population grows from around 254 million to six billion people between the years 1000 and 2000.
1800 (19th Century)
Between 1849 and 1905, the most prolonged period of drought conditions in 300 years occurred in Arizona.
1700 (18th Century)
"Little Ice Age" chills much of Europe, with glaciers growing in the Alps threatening some mountain communities and shortening growing seasons throughout Europe.
1600 (17th Century)
Slave trade, plantations and global commerce contribute to changes in land cover, influencing regional climate. Atmospheric CO2 levels are 6% below average Holocene level.
1500 (16th Century)
Severe multi-decadal "mega-drought" hits American southwest, severely impacting native peoples who had only recently been invaded by Spanish conquistadors. "Lost Colony" Drought also effects settlement of Jamestown, VA, 1587-89.
1400 (15th Century)
Sailors from Europe and the Middle East learn to navigate the world's oceans using seasonal wind patterns later called "trade winds". The "Little Ice Age" begins to chill much of Europe.
1300 (14th Century)
Possibily linked to wetter, colder climates, Bubonic plague kills up to 20,000 people a day in Cairo. Europe also hard hit. Empires thrive in Mali, Java and Uzbekistan. Minimum of solar activity during 14th Century.
1200 (13th Century)
1259- Evidence of major volcanic event-- likely the largest during entire Holocene-- found in ice cores on both poles.
Great Drought (1276-1299) in American Southwest found in tree ring data impacted Ancient Pueblo and other native cultures.
1100 (12th Century)
Called "The Century of the Axe" by some historians because of the ambitious building efforts and clearing of woods for agriculture in Europe and elsewhere. Changes in land cover eventually contribute to changes in regional climate.
1000 (11th Century)
~1000- Leif Eriksson, returning to Viking settlements in Greenland from Europe, is blown off course and lands on the North American continent. Medieval Warm Period in Europe begins around 1000 and lasts until approximately 1350 AD.

The figure to the left shows the amount of ice melting from an Ice Cap in what is now northern Canada. Reflecting the warming that has occurred after the most recent ice age, this graph provides a snapshot of the range of variability as measured by melting ice caps that can occur during a time span of 10,000 years. Note that the melting that has occurred during the 20th Century is greater than almost all periods for ~4000 years. (See dashed line).
Scientists looking for long-term patterns of 1,000 years or more in climate and environmental change use a variety of paleo proxies such as tree rings, and cores taken from ice caps and sediment layers from the ocean or lakes to glean data that can provide insight into why climate can abruptly shift in less than a decade.
The 1000-year increments below present climate-affected human activities during this period.
1000 years ago
Storage of rye results in occasional growth of a toxic fungal infection (ergot) known as St. Anthony’s fire that can cause hallucinations.
Chinese develop gunpowder.
2000 years ago
Spice trade becomes important. European sailors discover how to use monsoon patterns to their advantage.
3000 years ago
Peanuts grown in Peru. Iron age in Europe and Middle East.
Phoenicians sail in the Mediterranean while Polynesians sail the Pacific.
Iron Age begins around 2650 years ago.
4000 years ago
Bronze smelted in Middle East, combining copper and tin, beginning the Bronze Age.
Olives, peaches and apricots cultivated in Eastern Mediterranean.
Drought around 2000 B.C. may have contributed to the collapse of the Akkadia civilization in Mesopotamia, which is regarded as the world's first empire. Evidence of drought also found along Nile in Egypt, Indus in India and in the Great Basin of western North America.
5000 years ago
Five sacred crops in ancient China: soybeans, rice, wheat, barley and millet.
Sorghum used in sub-Saharan Africa.
Egypt unified under one Pharaoh.
6000 years ago
Neolithic (New Stone Age) period, with farming and elaborate stone houses built in Britain.
Cotton grown in Pakistan, cultivated grapes in Afghanistan.
7000 years ago
Squashes, beans, chili peppers and an early type of corn (Zea mays) cultivated in Meso-America.
Domesticated rice used in China
8000 years ago
Lentils, fava beans and chick peas become part of eastern Mediterranean, with Chili pepper and Lima beans being used in Peruvian highlands.
Rising sea levels of Mediterranean Sea floods into the basin that now forms the Black Sea around 7,600 years ago. (See Climate History 10,000 years)
Copper first smelted around this time period.
9000 years ago
During Mesolithic period, semi-permanent houses are used and boats are built for transport and fishing.
Flax in Syria and Turkey for clothing (linen) and oil.
Abrupt climate change causes much of the planet to become cooler and drier around 8,200 years ago.
10,000 years ago
-First evidence of plant domestication.
Wheat and barley developed in Near East. Barley becomes a daily food staple.
-An estimated 5 million people inhabit the entire planet.
Scientists have become increasingly aware of multi-millennial scale orbital cycles of precession, eccentricity, and obliquity which can play an important role in the rise and fall of ice ages. During the past 100,000 years ago, human beings—Homo sapiens —have developed from our hominid ancestors, adapting to rugged climates, such as in Europe 40,000 years ago.
Note: The Last Ice Age cycle lasted from roughly 60,000 to 20,000 years before present, with Ice-Age cycles occurring since 2.6 Million years ago to the present.
10,000 years before present (BP)
Beginning of Holocene. Large mammals including saber-toothed cats, mammoths, and mastodons become extinct. Neolithic period with beginning of agriculture and end of Ice Ages.
An estimated 5 million Homo sapiens inhabit planet Earth.
20,000 BP
Abrupt cooling about 15,000 years ago gives way to abrupt warming at the end of the Younger Dryas period some 11,600 years ago, with a climatic ripple effect impacting habitats around the world.
Gray wolves in East Asia become domesticated about 15,000 years ago, with all modern dogs evolving from them.
20,000 years ago, global mean temperature 4 degrees C cooler than today, although the North Atlantic was 14 degrees C cooler. Lower sea level allows large-scale migrations of people into the Americas.
30,000 BP
Homo sapiens thrive in cold European climate. Homo neanderthalensis (Neandertals) become extinct, with last fossil evidence dated 28,000 years ago in Portugal.
40,000 BP
Plethora of stone and bone tools along with cave paintings and other artwork in Europe. Homo sapiens use bone, ivory, antlers, and shells to make tools while Neandertals only use stone to make tools.
50,000 BP
Cave dwellers leave evidence of seeds of wild dates and nuts including chestnuts, walnuts, pine nuts, and acorns in Shanidar Cave of Northern Iraq.
Many large megafauna in Australia including large kangaroos, wombats and emu-like ducks, become extinct, possibly due to human hunting and use of fire.
60,000 BP
Homo sapiens enter Australia and begin to use fire, altering the existing flora and fauna.
During warming period (55-45K BP) mammoths roam central Sweden.
70,000 BP 80,000 BP 90,000 BP
Recent discoveries in caves along South African coast dating to 70,000 years before present suggest people using bone tools and living on fish and mammals in the region.
Major eruption of Mount Toba 73,000 years ago in modern day Sumatra impacts global climate system.
100,000 BP
Diet of Homo sapiens includes fish and seafoods as last Ice Age impacts Northern Hemisphere. Some scientists theorize that fish oil was key to the growth of the brain of Homo sapiens Evidence suggests that no other hominids such as Neanderthals ate fish. (See Broadhurst, 2001) Neandertals well established in Europe since at least 300,000 years before present.
![]() Principle 4e
Principle 4e
The Average Temperature is Warmer Now Than it has Been in 1,300 Years
Because records of temperature and precipitation using thermometers, rain gauges and the like have only been used for a few centuries, scientists need some source of reliable climate data before there were thermometers. So scientists use something called a “proxy.”
A proxy is a type of substitute (like a substitute teacher—it’s there when the original can’t be). Fortunately there are lots of climate proxies that allow scientists to extend the study of climate back thousands and even hundreds of thousands of years. Examples include: ice cores, ancient pollen, tree rings, boreholes, corals, lake and ocean sediments, and cave formations.
Read more…
The Average Temperature is Warmer Now Than it has Been in 1,300 Years
Because records of temperature and precipitation using thermometers, rain gauges and the like have only been used for a few centuries, scientists need some source of reliable climate data before there were thermometers. So scientists use something called a “proxy.”
A proxy is a type of substitute (like a substitute teacher—it’s there when the original can’t be). Fortunately there are lots of climate proxies that allow scientists to extend the study of climate back thousands and even hundreds of thousands of years. Examples include: ice cores, ancient pollen, tree rings, boreholes, corals, lake and ocean sediments, and cave formations.
An interesting example of a proxy used in Montana are packrat middens. Packrat middens are clumps of vegetation, insects, remains of vertebrates, and other materials cemented together by crystallized packrat urine (they call it amberat). These rock-hard deposits can be more than 20,000 years old and hold all kinds of clues to past climates.
From proxies, scientists are able to reconstruct past climates quite accurately, and, on occasion, they show abrupt changes in climate. 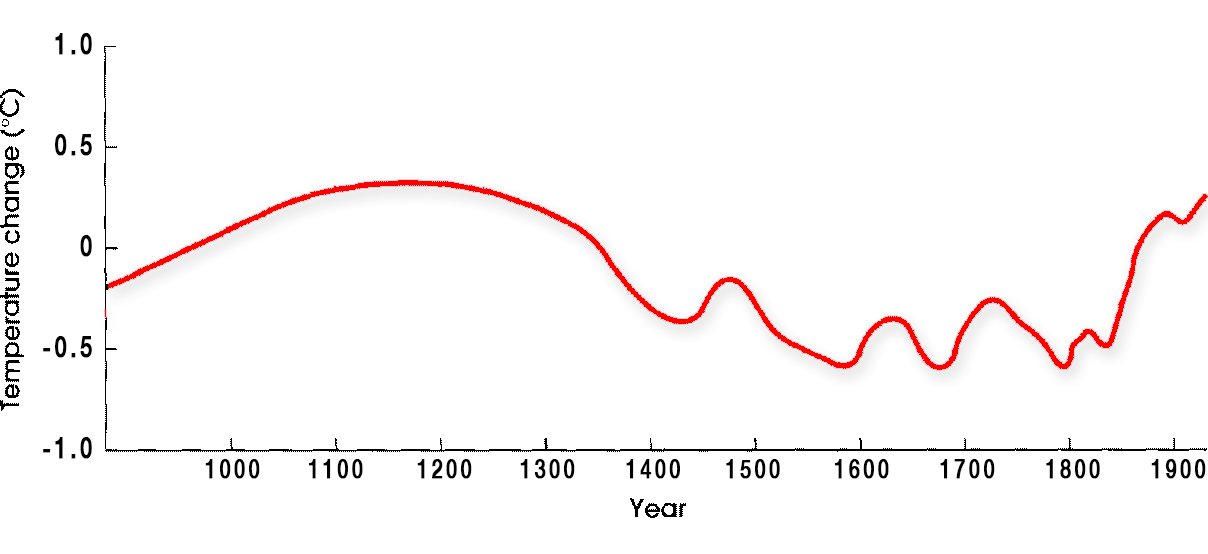 Based on evidence from all of these different types of proxies along with scientific observations made around the world, we know the Earth’s average temperature is warmer today now it has been for at least the past 1,300 years. Average temperatures have increased markedly in the past 50 years, especially in the North Polar Region. In fact, the Earth has been warming since when industrial-era fossil fuel emissions started nearly 200 years ago.
Based on evidence from all of these different types of proxies along with scientific observations made around the world, we know the Earth’s average temperature is warmer today now it has been for at least the past 1,300 years. Average temperatures have increased markedly in the past 50 years, especially in the North Polar Region. In fact, the Earth has been warming since when industrial-era fossil fuel emissions started nearly 200 years ago.
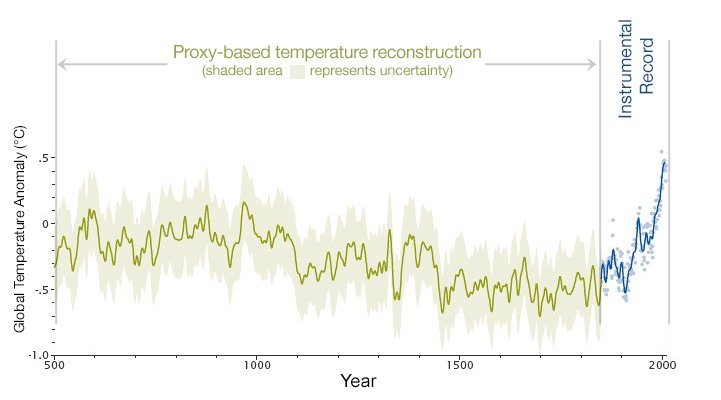
The Rate of Change is Different Too: NASA says Earth is warming at a pace 'unprecedented in 1,000 years'
NASA says that records of temperature taken via analysis of ice cores and sediments, suggest that the warming of recent decades is out of step with any period over the past millennium. “In the last 30 years we’ve really moved into exceptional territory,” Gavin Schmidt, director of Nasa’s Goddard Institute for Space Studies, said. “It’s unprecedented in 1,000 years. There’s no period that has the trend seen in the 20th century (in terms of how fast temperatures are increasing).”
Paleoclimatology: How Can We Infer Past Climates?
Created by Monica Bruckner, Montana State University.
What is Paleoclimatology?

Past climate can be reconstructed using a combination of different types of proxy records. These records can then be integrated with observations of Earth's modern climate and placed into a computer model to infer past as well as predict future climate.
How Are Organisms Used As Proxies?

Researchers may also use foram and diatom population dynamics to infer past climate. Relative abundance as well as species composition in particular areas may indicate environmental conditions. Typically, warmer weather will cause organisms to proliferate. In addition, since each species has a particular set of ideal growing conditions, species composition at a particular site at a particular time may indicate past environmental conditions.
How Are Other Proxies Used?
Combinations of proxy data are generally used to reconstruct records for past climate. In addition to forams and diatoms, common proxies and their respective analytical methods include:



What Causes Climate Change?
The causes of climate change are complex. There are several major factors that can effect the climate system, including:
- Changes in solar output
- Changes in Earth's orbit
- Changes in the distribution of continents
- Changes in atmospheric content of greenhouse gases.

It is important to consider scale when interpreting climate change through time. Four major time scales are generally considered, which include:
- Long term- Hundreds of millions of years;
- Medium term- One million years;
- Short term- ~160,000 years;
- Modern period- Hundreds of years.
Time scale affects interpretations of climate change. Climate has both long term trends and short term variability. In looking at longer time scales, major shifts in climate such as the ice ages are easily recognizable, and viewing a long-term data set can provide the observer with a sense of the "big picture" of the climatic trends. Short term variations, like a colder than average month, can exist within longer term patterns such as the warming trend over the past 1000 years. The coexistence of short and long term trends occuring simultaneously through time complicates our ability to unravel climate change.
Why is Paleoclimatology Important?
The science of paleoclimatology is important for past, contemporary, and future issues. Understanding past climate helps us to explain how current ecosystems came to be. For example, climate typically controls what types of vegetation grow in a particular area. Furthermore, paleoclimatology provides data that we can use to model and predict both current and future climate change scenarios. Computer models can be used to study the potential effect of increased atmospheric carbon dioxide on climate.
With a system as complex as Earth's climate, it is a daunting task for scientists to be able to make projections about future climate changes and how it may affect the distribution of plants and animals. However, paleoclimate data are used as a foundation for climate scientists by providing crucial information such as rates of past climate change and how vegetation and animal populations responded to the change. Computer models can be used predict different future climate patterns, and paleoclimate data provides a useful framework from which to base these models. For more information about climate proxies and climate change, see the collections of web-based materials below.
Climate Proxy Collections



Climate change now has a start date
Source: http://www.climatechangenews.com/2016/08/24/climate-change-now-has-a-start-date/
Researchers have pinpointed the beginning of global warming to a couple of decades in mid-1800s, showing earth’s sensitivity to small atmospheric changes
Opening of the Liverpool and Manchester Railway in 1830, the date scientists now believe emissions from the UK's industrial revolution began to warm the planet. Source: Wikimedia Commons
By Karl Mathiesen
On December 4, 1830, the Planet chugged out of Liverpool on its maiden trip to the great manufacturing centre of Manchester.
Shovelled full of coal, the steam locomotive was hauling freight along the world’s first intercity rail route – a major advance in the industrialisation of the globe.
It was around this moment, scientists have discovered, that our own planet began to go off the rails.
Using 2,000 years of paleoclimate data or proxies – the earth’s historical temperature measured from natural sources such as the growth bands of corals and trees, ice cores and the amount of pollen trapped in sediment layers – a global team of researchers lead by Australian National University associate professor Nerilie Abram, have pinpointed the moment when the earth’s temperature began to rise because of human greenhouse gas emissions to between 1830 and 1850.
Abram said the findings, published on Wednesday in the journal Nature, were “extraordinary” and had implications for our understanding of the sensitivity of the globe to even tiny increases of carbon in the atmosphere.
A scientist extracts coral cores at Rowley Shoals, west of Broome in Western Australia.
Source: Eric Matson, Australian Institute of Marine Science
Study co-author, Dr Helen McGregor, from the University of Wollongong said: “The early onset of warming detected in this study indicates the earth’s climate did respond in a rapid and measurable way to even the small increase in carbon emissions during the start of the industrial age.”
Abrams said the increase in atmospheric carbon between the onset of warming and the end of the 1800s was “small”, around 15 parts per million. But even this raised the temperature by a few tenths of a degree. The increase since 1900 has been more than 100 parts per million.
Paleoclimate temperature records were most famously analysed in the 1990s by US scientist Michael Mann to produce the “hockey stick” graph, which shocked the world with its dramatic depiction of the rapid recent rise in temperature after a millennia of relative stability.
400ppm: The milestone that puts Earth in the “danger zone”
But these natural almanacs have never so accurately calculated the beginning of human-induced warming.
In a paleoclimatological first, Abram’s study incorporated not only land based sources like tree rings, but measured marine temperatures as well. The scientists found the land of the northern hemisphere and seas of the tropics began warming at roughly the same speed around 1830.
“Seeing that parts of the oceans are a very responsive part of the climate system is a new and very interesting bit of information,” said Abram. Particularly because those oceans contain some of the most climate sensitive ecosystems, coral reefs. The southern hemisphere was around 50 years behind. This was likely the result of cooling currents in the huge southern oceans.
This regional variability also allowed Abram’s team to chart the different stages of “emergence” around the globe. That is the point at which the average temperature has increased so much that it exceeds even extreme natural fluctuations.
“In the tropical oceans and the Arctic in particular, 180 years of warming has already caused the average climate to emerge above the range of variability that was normal in the centuries prior to the Industrial Revolution,” said Abram. The Antarctic, however, remains stubbornly un-warmed to this day.
Because of the huge technological leaps and enormous new wealth that drove projects like the Liverpool and Manchester Railway, early industrial emissions were dominated by the United Kingdom.
According to the World Resource Institute, by 1850 the furnaces of the British industrial revolution had belched 122.6 million tons of climate-warming carbon dioxide into the atmosphere.
This cumulative total was twice as much as the rest of the world combined had emitted to that point – most of it coming from Britain’s great rivals, France, the US and Germany. Today, Britain emits around 500 million tonnes every single year.
The findings shift our understanding of exactly what is normal, because instrumental records of the global temperature only reliably go back as far as 1880.
Using that baseline, Nasa has determined that the first six months of 2016 were 1.3C warmer than normal. But Abram says the world had already warmed a few tenths of a degree by 1880, pushing the world beyond the 1.5C limit already.
“That’s important for conversations that we are having at the moment about trying to limit warming to 1.5C. We are getting scarily close to that already, but that’s when we are talking about the baseline being in the 1880s-1900. So we don’t yet have the full picture,” she said.
![]() Principle 4f
Principle 4f
We Have Known for a Long Time How the Greenhouse Effect Works
A generation after John Tyndall developed the theory and conducted observations that led to the concept of the greenhouse effect, Svante Arrehenius (1859-1927) in Sweden made calculations on the influence of carbon dioxide in the air upon the temperature of the ground. His interest was motivated by the observation that burning coal—which was widespread across Europe—added carbon dioxide (CO2) to the air. His calculations suggested that adding CO2 could cause the planet to warm, in effect amplifying the effect of greenhouse gases already naturally in the atmosphere.
Read more…
That means the study of the greenhouse effect predates manned flight (1903), and the discoveries of penicillin (1928) and of Neptune (1846). Below are some paraphrased portions of a climate change timeline that can be found at Spencer Weart’s informative “The Discovery of Global Warming” website:
1824
Joseph Fourier calculated the Earth would be colder without an atmosphere.
1847
George Perkins Marsh, the designer of the Washington Monument and the Smithsonian Institution, delivered an address before the Agricultural Society of Rutland County, Vermont, that warned of climate change: “…it is certain that climate itself has in many instances been gradually changed and ameliorated or deteriorated by human action. The draining of swamps and the clearing of forests perceptibly effect the evaporation from the earth [...] The same causes modify the condition of the atmosphere and the power of the surface to reflect, absorb and radiate the rays of the sun, and consequently influence the distribution of light and heat, and the force and direction of the winds. Within narrow limits too, domestic fires and artificial structures create and diffuse increased warmth…” He was talking about concepts familiar to us now as the urban heat island effect and the greenhouse effect.
1859
John Tyndall discovers some gases block infrared radiation, and proposes changes in their concentration could bring climate change.
1896
Svante Arrhenius first calculates global warming from human-produced CO2 emissions.
1897
Thomas Chrowder Chamberlin produces a global carbon exchange model including feedbacks.
1938
Guy Stewart Callendar says CO2 greenhouse global warming is happening.
1956
Gilbert Plass says adding CO2 to the atmosphere has a major effect on the radiation balance.
1957
Roger Revelle finds that CO2 produced by humans will not be readily absorbed by the oceans.
1958
Venus’ greenhouse effect (which raises the atmosphere’s temperature above the boiling point of water) is observed by telescope.
1960
Charles Keeling detects an annual rise in the Earth’s atmospheric CO2.
1968
Studies say Antarctic ice sheets may collapse, which would raise sea levels big time.
The father of climate change
Ian Sample looks at how the study of the climate has moved from being a relatively minor branch of science to one that now dominates most others, thanks largely to the work of one man
Source: http://www.theguardian.com/environment/2005/jun/30/climatechange.climatechangeenvironment2
Behind the treelined embankment that borders the campus of Stockholm University lies building 92E, a red brick villa as big as a fire station, its back turned to Roslagsvägen, the main artery linking the capital city with Norrtälje 70km away.
What few markings there are on the building suggest nothing of its history. A sign above the entrance identifies it as Cafe Bojan, a student canteen, and a few shirtless students on a bench in the morning sun recall it as nothing more.
At the end of the 19th century, building 92E was the home and laboratory of Svante Arrhenius, a chemist who became Sweden's first Nobel prizewinner. He was destined to have a bigger impact than he could have imagined, far beyond his mainstream work. Unwittingly, he uncovered secrets of the Earth's atmosphere and in doing so triggered research into what many see as the biggest threat to modern humans. He is arguably the father of climate change science.
That title would be a surprise, even to him. The son of a land surveyor, Arrhenius thrived at school, showing a particular aptitude for arithmetic, but his diversity of thought and penchant for maverick theories dealt him a hefty blow at university. His PhD research, which he began at Uppsala University to the north of Stockholm, focused on the conductivity of electrolytes, but the ideas he put forward in his thesis baffled his professors and he was awarded the lowest possible pass grade. At once, any hopes of staying on at Uppsala were destroyed, and Arrhenius embarked on a tour of European laboratories before landing a job in Stockholm several years later.
Arrhenius became interested in a debate occupying the scientific community, namely the cause of the ice ages. Could it be, he wondered, that vast swings in the levels of atmospheric CO2, lasting tens of millions of years, were the trigger?
The link between CO2 and the Earth's temperature had been made years beforehand. It was the French scientist Joseph Fourier who first realised that certain atmospheric gases shrouded the planet like a bell jar, transparent to sunlight, but absorbing to infrared rays. It means the atmosphere is heated from above and below: first, by sunlight as it shines through and second by the infrared the Earth emits as it cools overnight.
Arrhenius set himself the task of working out just how much water and CO2 in the atmosphere warmed the planet. From others' work, he knew that CO2 was only part of the process. While CO2 and other gases trapped infrared radiation and so heated the atmosphere, warmer air holds more water vapour, itself the most potent contributor to the greenhouse effect. So, if atmospheric CO2 levels increased, water vapour would ensure the warming effect was seriously magnified.
What followed was a year doing what Arrhenius described as "tedious calculations". His starting point was a set of readings taken by US astronomer Samuel Langley, who had tried to work out how much heat the Earth received from the full moon. Arrhenius used the data with figures of global temperatures to work out how much of the incoming radiation was absorbed by CO2 and water vapour, and so heated the atmosphere.
Between 10,000 and 100,000 calculations later, Arrhenius had some rough, but useful, results that he published in 1896. If CO2 levels halved, he concluded, the the Earth's surface temperature would fall by 4-5C. There was a flipside to his calculations: doubling CO2 levels would trigger a rise of about 5-6C.
Beyond the argument over ice ages it wasn't lost on Arrhenius that human activity, in the form of widespread burning of coal, was pumping atmospheric CO2 above the natural levels that help make the Earth habitable. Almost as a passing comment, he estimated that coal burning would drive a steady rise in CO2 levels of about 50% in 3,000 years, a prospect he found entirely rosey. At a lecture that same year, he declared: "We would then have some right to indulge in the pleasant belief that our descendants, albeit after many generations, might live under a milder sky and in less barren surroundings than is our lot at present."
As the first to put hard figures on the greenhouse effect, it's unsurprising Arrhenius's estimates weren't spot on. He thought it would take millenia to see a 50% rise in CO2 - but modern measurements show a 30% rise during the 20th century alone. He thought a doubling of CO2 would raise temperatures by 5-6C. Scientists now say 2-3C is more likely.
Over the next decades, his work was criticised, backed up and criticised again. Many disregarded his conclusions, pointing to his simplification of the climate and how he failed to account for changes in cloud cover and humidity. The oceans would absorb any extra CO2 pumped into the atmosphere, and any remainder would be absorbed by plant life, leading to a more lush landscape, sceptics argued.
In 1938, nine years after Arrhenius had died a Nobel prizewinner for his work on ionic solutions, English engineer Guy Callendar gave the greenhouse theory a boost. An expert on steam technology, he took up meteorology as a sideline and became interested in suggestions of a warming trend. Callendar pieced together temperature measurements from the 19th century onwards and saw an appreciable rise. He went on to check CO2 over the same period and discovered levels had increased about 10% in 100 years. The warming was probably due to the higher levels of CO2.
The existence of an increasing greenhouse effect was hotly debated until the postwar funding of the 1950s kicked in and researchers began to get firm data. In 1956, physicist Gilbert Plass confirmed adding CO2 to the atmosphere would increase infrared radiation absorbed, adding that industrialisation would raise the Earth's temperature by just over 1C per century. By the end of the 1950s, Plass and other scientists in the US started warning government officials that greenhouse warming might become a serious issue in the future.
Unwittingly, the US especially had already started monitoring what many believed were the direct effects of a warming world. Submarines operating in the Arctic Circle took accurate readings of the thickness of the ice sheets above them. When the Pentagon released the data nearly 40 years later, it revealed a startling melting of the ice, on average a 40% thinning of 1.3m since 1953.
In the 1960s, researchers at Scripps Institution of Oceanography in San Diego took on the testing challenge of taking a vast number of measurements of atmospheric CO2. The aim was to establish a baseline level with which future readings in a decade or so could be compared.
Charles Keeling spent two years taking measurements in Antarctica and above the Mauna Loa volcano in Hawaii but reported that even in this short period, CO2 levels had risen. He concluded that the oceans weren't absorbing greenhouse gases being pumped out by industry. Instead, emissions were driving levels of CO2 higher. "It was a seminal discovery. For the first time, scientists knew that the oceans weren't going to absorb all this carbon dioxide," says Mike Hulme at the Tyndall Centre for climate change research at the University of East Anglia.
Still, few saw the greenhouse effect and the warming it would bring as being a problem. At the time, computer models were suggesting modest increases, perhaps 2C in hundreds of years.
By the 1980s, climate change had become a megascience, attracting scientists from diverse fields, each attacking the problem from a different angle. One technique was especially useful. Deep cores of ice cut from Greenland and elsewhere held pockets of air dating back hundreds of thousands of years. By analysing the trapped air, scientists worked out CO2 levels in the atmosphere during past ice ages. In 1987, a core cut from central Antarctica showed that in the previous 400,000 years, CO2 had dropped to 180 parts per million (ppm) during the most extreme glacial periods and climbed as high as 280ppm in warmer times, but not once had been higher. In the outside air, CO2 was measured at 350ppm, unprecedented for nearly half a million years.
To mainstream scientists, evidence that warming was down to human activity was becoming too big to ignore. While scientists uncovered evidence for the greenhouse effect and warming it was producing, others pointed to different processes impacting on global climate. Volcanos, for example, blast millions of tonnes of sulphur dioxide into the atmosphere that form aerosol particles which reflect sunlight back into space. The 1991 eruption of Mount Pinatubo in the Phillipines sent about 20m tonnes of the gas into the atmosphere, leading to a global cooling of around 0.5C a year later. Scientists now believe that the warming experienced in the early 20th century can largely be explained by the lack of volcanic activity.
Variations in the sun's intensity have also been fingered as a driver of climate change. According to Joanna Haigh at Imperial College London, about a third of the warming since 1850 can be explained by solar activity. The identification of disparate contributors to warming has been seized upon by a minority who claim global warming is driven far more by nature than human activity, and the ensuing controversy is still not settled.
By 1988, the United Nations had established the Intergovernmental Panel on Climate Change to review relevant research. The panel's latest estimate points to a warming of 1.4-5.8C by 2100, depending on what strategies, if any, are adopted to curb emissions. The 20th century saw a rise in temperature of 0.6C, about half of which occured since 1970.
Arguably the most concerted effort to cut global emissions has been triggered by the Kyoto Protocol. Since ratification began in 1997, more than 100 countries have adopted the protocol, which for the first time committed them to cutting emissions of six greenhouse gases.
Now, barely a week goes by without a major study on climate change. A flurry of papers started the year with warnings that the Gulf Stream would grind to a halt, ski resorts would move to higher altitudes and Antarctic glaciers were melting fast. More than 100 years after Arrhenius set out to discover why the world fell into periodic ice ages, the scientist has become a pillar of the megascience that is global warming research.
Back in Stockholm' meteorology department, Erland Kallen is musing about progress since Arrhenius first set about his calculations. "Even when I came to this field 20 years ago, I was very sceptical about global warming. There were too many uncertainties I just couldn't see how anyone could say anything sensible about it. Now, I struggle to see what other explanation there could be."
Timeline
1898
Swedish scientist, Svante Arrherius, puts forward the theory of the greenhouse effect and calculates that doubling of carbon dioxide in the atmosphere will increase temperatures by 5°C to 6°C
1956
US weapons researcher Gilbert Plass pursues climate research in his free time and analyzes how carbon dioxide traps heat. He announces that climate change could be a severe problem to future generations.
1979
The First World Climate Conference, sponsored by the World Meteorological Organization, is held in Geneva, Switzerland. Extremeweather events earlier during the decade had focussed public attention on climate.
1982
Ice cores from the Greenland ice sheet show dramatic temperature oscillations in a single century from the past, an extremely short period for climate change. Scientists also call 1981 the warmest year on record.
1997
The Kyoto Protocol is negotiated to reduce greenhouse gas emissions 5.2% below 1990 levels by 2012 in the developed countries including the former communist bloc.
2005
Kyoto Protocol enters into legal force on Feb 16. The treaty was ratified by more than 140 countries. Concentration of carbon dioxide now stands at 372 parts per million, higher than at any time in at least the past 420,000 years.
![]() Principle 4g
Principle 4g
Carbon Dioxide Stays in the Atmosphere for a Century or Longer
Natural processes that remove carbon dioxide from the atmosphere operate very slowly compared to the processes that are now adding it to the atmosphere. Thus, carbon dioxide introduced into the atmosphere today will remain there for a century or longer. Other greenhouse gases, including some created by humans, will remain in the atmosphere for thousands of years.
Moving greenhouse gases from the atmosphere into terrestrial and oceanic sinks is a process so slow it is difficult to comprehend. CO2 can be in the atmosphere for thousands of years. The geologic processes trapping atmospheric carbon into sedimentary rocks, like limestone, take hundreds of millions of years. Read more…
Computer models estimate that warming generated by current carbon dioxide emissions will persist for as long as 1,000 years after emissions stop. While the removal of atmospheric carbon dioxide does decrease warming, it takes a long time for the heat to dissipate and be absorbed by the oceans. As a result, atmospheric temperatures will not drop significantly for a long period of time, even after we slow our output of carbon dioxide.
Take a bite of dinner, a breath, or a drive in a car — you are part of the carbon cycle — moving carbon from one reservoir to another.
Source: NOAA (Featured in ESRL's Carbon Cycle Toolkit)
Carbon is the chemical backbone of life on Earth, a key element in many important processes. Carbon compounds help to regulate the Earth’s temperature, make up the food that sustains us, and provide a major source of the energy to fuel our global economy. Most of Earth’s carbon is stored in rocks and sediments, while the rest is located in the ocean, atmosphere, and in living organisms - these are the reservoirs through which carbon cycles.

Carbon moves from one storage reservoir to another through a variety of mechanisms. One example is the movement of carbon through the food chain. Plants move carbon from the atmosphere into the biosphere through photosynthesis: they take in carbon dioxide and use energy from the sun to chemically combine it with hydrogen and oxygen to create sugar molecules. Animals that eat the plant can digest the sugar molecules to get energy for their bodies. Respiration, excretion, and decomposition release the carbon back into the atmosphere or soil continuing the cycle.
The ocean plays a critical role in the storage of carbon, as it holds about 50 times more carbon than the atmosphere. Two-way carbon exchange can occur quickly between the ocean’s surface waters and the atmosphere, but carbon may also be stored for centuries at the deepest ocean depths.
Rocks such as limestone and fossil fuels such as coal and oil are storage reservoirs that contain carbon from plants and animals that lived millions of years ago. When these organisms died, slow geologic processes trapped their carbon and transformed it into these natural resources. Processes such as erosion release this carbon back into the atmosphere very slowly while volcanic activity can release it very quickly. Burning of fossil fuels in cars or power plants is another way this carbon can be released into the atmospheric reservoir quickly.
Changes to the Carbon Cycle
The increasing human population and their activities have a tremendous impact on the carbon cycle. Burning of fossil fuels, changes in land use, and the use of limestone to make concrete all transfer significant quantities of carbon into the atmosphere. As a result the amount of carbon dioxide (CO2) in the atmosphere is rapidly rising and is already significantly greater than at any time in the last 800,000 years. This increase of CO2 is affecting our ocean as it absorbs much of the CO2 that is released from burning fossil fuels. This extra CO2 is lowering the ocean’s pH, this process is called ocean acidification and interferes with the ability of marine organisms (such as corals) to build their shells and skeletons.
![]() Principle 4h
Principle 4h
Local Relevance
A Different Climate, A Different World: During the Last Ice Age, the Pacific Northwest Experienced Enormous Floods
An Introduction to the Ice Age Floods
During the last Ice Age (18,000 to 12,000 years ago), and in multiple previous Ice Ages, cataclysmic floods inundated portions of the Pacific Northwest from Glacial Lake Missoula, pluvial Lake Bonneville, and perhaps from subglacial outbursts. Glacial Lake Missoula was a body of water as large as some of the USA’s Great Lakes. This lake formed from glacial meltwater that was dammed by a lobe of the Canadian ice sheet. Perhaps every 40 to 140 years, the waters of this huge lake forced its way past the ice dam, inundating parts of the Pacific Northwest. Eventually, the ice receded northward far enough that the dam did not reform, and the flooding episodes ceased. These floods are a remarkable part of North American natural heritage. They have profoundly affected the geography and ways of life in the region, but have until recently remained largely unknown to the general public.
The Ice Age Floods Story – Briefly
During the most recent episode of major ice-sheet expansion, between about 18,000 and 13,000 years ago, a lobe of the Cordilleran ice sheet advanced into the Idaho Panhandle to the area that is now occupied by Lake Pend Oreille, thus blocking the Clark Fork River drainage and causing Glacial Lake Missoula to form. At its largest, the lake was deeper than 2,000 feet deep at the dam and held over 500 cubic miles of water—as much as Lake Erie and Lake Ontario combined. The ice dam, however, was subject to repeated failure.
When the dam broke, a towering mass of water and ice was released and swept across parts of Idaho, Washington, and Oregon on its way to the ocean. The peak rate of flow was ten times the combined flow of all the rivers of the world. The huge lake may have emptied in as little as two or three days. Over a period of years the glacier would advance, once again blocking the river, and the dam and the lake would form again. This process was repeated scores of times, until the ice sheet ceased its advance and receded to the north at the end of the Ice Age. It is assumed that the same processes would have occurred earlier during other glacial advances throughout the Ice Age, although most of the evidence for the earlier events may have been removed by the flooding that occurred during the last glacial advance.
Areal Scenario Map of the Ice Age Floods.
Along the floodwaters’ course, more than 50 cubic miles of earth and rock were removed, and some of this was transported and then deposited as new landforms. The floods built gravel bars as tall as 400 feet and moved boulders weighing many tons as if they were pebbles. Some of the eroded material was deposited along the path of the floods, but most of the eroded material was carried out onto the floor of the Pacific Ocean, where extensive deposits of flood sediment have recently been identified hundreds of miles from the current mouth of the Columbia River.
In the following years the account was refined, as evidence of more than one flood was discovered. It is now established that there were large numbers of Ice Age floods that swept across the US Northwest, and some of them were among the largest and most powerful floods that have ever occurred on Earth.
These floods are a remarkable part of North American natural heritage. They have profoundly affected the geography and ways of life in the region, but have until recently remained largely unknown to the general public.
How the “Story” Began
Early settlers in eastern Washington State recognized that the landscapes were different than those seen in other places but were not sure why. A closer examination of the features in the Basin led one geologist, J. Harlen Bretz, to propose that only a sudden cataclysmic flood, on a scale never before considered possible, could account for the phenomenal size and distinctive characteristics of the landforms. This radical idea was not well received by fellow geologists, and a long-running scientific dispute followed. Ultimately his extensive field work, plus additional research by others, conclusively established that many extraordinarily huge and powerful Ice Age floods had shaped the region.
It was in 1923 that Bretz published the first in a series of scientific papers in which he proposed that the severely eroded Channeled Scabland, Dry Falls, and other immense geologic features had been formed by huge, powerful floods that had swept through the Columbia Basin during the Ice Age.
Despite his peers’ doubt and opposition, he resolutely maintained that direct examination of the geologic evidence could lead only to that conclusion. But Bretz was unable to identify the source or cause of such catastrophic flooding.
Earlier, in 1910, another geologist, Joseph T. Pardee, had described evidence of a great ice dammed lake, Glacial Lake Missoula, that had formed during the Ice Age in northwestern Montana. However, Bretz didn’t see the connection between the glacial lake in Montana and the features he described in eastern Washington. Then, in 1940, Pardee reported on his discovery of giant ripple marks, 50 feet high and 200-500 feet apart, that had formed on the floor of Glacial Lake Missoula. These huge, current-related features, along with other newly-found landforms, dramatically confirmed that the lake had suddenly emptied to the west, unleashing the tremendously powerful erosive forces that shaped many of the landforms found in the Columbia Basin. More research followed, and new perspectives became available from aerial photography. Among geologists, the concept of a catastrophic flood came to be accepted by the late 1950s.
Phenomena related to the Ice Age Floods
Recent research has found evidence that comparable floods occurred much earlier in the Ice Age in the Columbia Basin, as much as 1 to 2 million years ago. It has been determined that huge Ice Age glacial-outburst floods occurred in other parts of the world, as well. Even in our own times, similar but much smaller floods have occurred. Scientific study of the Ice Age Floods is contributing to the understanding of cyclical climate change and of very large and destructive contemporary floods on Earth. The Ice Age Floods have also been considered as an analog to understand geologic processes on Mars, where landforms strikingly similar to those in Eastern Washington exist. Could huge floods on this scale happen again? Although global warming may now be a serious concern, it is likely that long-term climate cycles will cause large ice sheets to return at some time in the distant future, and cataclysmic outburst floods will probably recur in this region.
![]() Principle 4i
Principle 4i
Misconceptions about this Principle
The Misconception
Isn’t it true that human-generated CO2 is just a tiny percent of total CO2 emissions and so cannot be responsible for climate change?
The misconception goes something like this: The oceans contain 37,400 billion tons of suspended carbon, land biomass has 2000-3000 billion tons. The atmosphere contains 720 billion tons and humans contribute only 6 billion tons additional load on this balance. The additional load by humans is incredibly small. A small shift in the balance between oceans and air would cause a much more severe rise in CO2 than anything we could produce.
The Science
Atmospheric CO2 is at its highest level in 10 to 15 million years due to the burning of fossil fuels. Human CO2 emissions have upset the natural balance of the carbon cycle.
CO2 in the atmosphere. NASA.
The science says: before the industrial revolution, the CO2 content in the air remained quite steady for thousands of years. Natural CO2 is not static, however. It is generated by natural processes, and absorbed by others. Read More…
Source: https://www.skepticalscience.com/human-co2-smaller-than-natural-emissions.htm

But consider what happens when more CO2 is released from outside of the natural carbon cycle – by burning fossil fuels. Although our output of 29 gigatons of CO2 is small compared to the 750 gigatons moving through the carbon cycle each year, it adds up because the land and ocean cannot absorb all of the extra CO2. About 40% of this additional CO2 is absorbed. The rest remains in the atmosphere, and as a consequence, atmospheric CO2 is at its highest level in 10 to 15 million years. (A natural change of 100ppm normally takes 5,000 to 20,000 years. The recent increase of 100ppm has taken just 120 years).
Human CO2 emissions upset the natural balance of the carbon cycle. Man-made CO2 in the atmosphere has increased by a third since the pre-industrial era, creating an artificial forcing of global temperatures which is warming the planet. While fossil-fuel derived CO2 is a very small component of the global carbon cycle, the extra CO2 is cumulative because the natural carbon exchange cannot absorb all the additional CO2.
The level of atmospheric CO2 is building up, the additional CO2 is being produced by burning fossil fuels, and that build up is accelerating.
Source: https://www.skepticalscience.com/human-co2-smaller-than-natural-emissions.htm
Principle 4
Check your Knowledge of this Principle
To pass this knowledge check you will need to have read the main paragraphs for each topic of the principle.